Arrange the atoms and/or ions in the following groups in order of decreasing size. a. 0, 0,
Question:
Arrange the atoms and/or ions in the following groups in order of decreasing size.
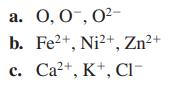
Transcribed Image Text:
a. 0, 0, 0²- b. Fe²+, Ni²+, Zn²+ c. Ca²+, K+, Cl-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a In this group all the species are oxygen ions To arrange ...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
2019A Company 1. The following information is taken from the records of 2019A Company for July: Purchases Direct materials Sales Salaries and wages: Selling and administrative salaries Direct...
-
40. Select the most acidic compound from the following. A. SO 2 B. Al 2 O 3 C. CaO D. PbO E. H 2 O 41. Select the most basic compound from the following. A. Bi 2 O 3 B. SiO 2 C. Cs 2 O D. Na 2 O E. H...
-
Arrange the following atoms in order of decreasing atomic radius: Na, Al, P, Cl, Mg.
-
You name the catastrophe, and JIT has been through it and survived. Toyota Motor Corporation has had its world-renowned JIT system tested by fire. The massive fire incinerated the main source of...
-
Is a current account deficit always associated with a strong real exchange rate (that is, one in which the currency is overvalued compared to the PPP prediction)?
-
The data presented in Table 6 represent the educational attainment of residents of the United States 25 years or older in 2013, based on data obtained from the U.S. Census Bureau. The data are in...
-
North Shore Architectural Stone, Inc., a company that installs limestone in residential and commercial buildings, agreed to supply and install limestone for a property owned by Joseph Vitacco. North...
-
Stevens Textiles's 2010 financial statements are shown below: Balance Sheet as of December 31, 2010 (Thousands of Dollars) a. Suppose 2011 sales are projected to increase by 15% over 2010 sales. Use...
-
The money supply process involves various factors, including the actions of commercial banks, the central bank's monetary policy, and the public's demand for money. My questions are: How do these...
-
Which of the following would you expect to be more favorable energetically? Explain. a. An H 2 molecule in which enough energy is added to excite one electron from the bonding to the antibonding MO...
-
Classify the bonding in each of the following molecules as ionic, polar covalent, or nonpolar covalent. a. H 2 b. K 3 P c. NaI d. SO 2 e. HF f. CCl 4 g. CF 4 h. K 2 S
-
Allan Co. had the following balances on December 31, 2015: The accountant for Allan has asked you to make an adjustment since $500 worth of janitorial services have just been performed for customers...
-
With reference to Graph B, at a price of $5, total revenue equals: Price $10- $5 O 20 40 60 80 100 Quantity Graph A Price Dinelastic $10 $5 D B 10 20 30 40 Quantity Graph B DElastic
-
Could you elucidate the concept of a relational database and elaborate on its function in facilitating customer communications within e-commerce enterprises?
-
4. Compare and contrast cost-only analysis and cost and effort analyses with regard to the cost that is included in the analysis. What effects would an underestimation of costs have? What effects...
-
what are the benefits of integrating artificial intelligence for demand forecasting
-
A convex lens has a focal length of 30 cm. If an object is placed 90 cm from the lens, where is the image? Enter your answer in centimeters. QUESTION 2 B. The image formed by the above lens is...
-
The realization principle determines when a business should recognize revenue. Listed next are three common business situations involving revenue. After each situation, we give two alternatives as to...
-
State whether each of the following will increase or decrease the power of a one-way between-subjects ANOVA. (a) The effect size increases. (b) Mean square error decreases. (c) Mean square between...
-
Calculate the pH of a 0.010-M solution of iodic acid (HIO 3 , K a = 0.17).
-
A solution is tested for pH and conductivity as pictured below: The solution contains one of the following substances: HCl, NaOH, NH 4 Cl, HCN, NH 3 , HF, or NaCN. If the solute concentration is...
-
When determining the pH of a weak acid solution, sometimes the 5% rule can be applied to simplify the math. At what K a values will a 1.0-M solution of a weak acid follow the 5% rule?
-
What narrative devices does the author utilize to maintain suspense and tension throughout the story, keeping the reader eagerly turning the pages?
-
Sandhill Construction Co. contracted to build a bridge for $11,700,000. Construction began in 2024 and was completed in 2025. Data relating to the construction are: 2024 2025 Costs incurred during...
-
The 3 0 - day T - bills have a current yield of 6 . 4 % with inflation premium at 3 . 5 % , liquidity premium at 1 . 2 5 % , maturity risk premium at 2 . 1 5 % , and default risk premium at 2 . 7 5 %...

Study smarter with the SolutionInn App


