Balance the following equations: a. Cr(s) + Sg(s) CrS3(s) b. NaHCO3(s) Heat, NaCO3(s) + CO(g) +
Question:
Balance the following equations:
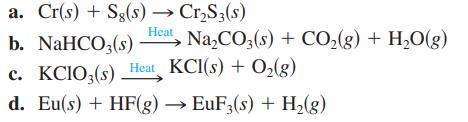
Transcribed Image Text:
a. Cr(s) + Sg(s) → Cr₂S3(s) b. NaHCO3(s) Heat, Na₂CO3(s) + CO₂(g) + H₂O(g) c. KCIO3(s) Heat KCl(s) + O₂(g) d. Eu(s) + HF(g) → EuF3(s) + H₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a Crs Sgs CrS38 This equation is already balanced Both sides have one chromium Cr atom and on...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) CH3COOH (aq) + KOH(aq)- .co.(aq) + NaO H (aq) - HNO3(aq ) + Ba(OH)2(aq )-
-
Balance the following equations and indicate whether they are combination, decomposition, or combustion reactions: (a) C3H6(g) + O2 (g) CO2 (g) + H2O(g) (b) NH4NO3(s) N2O(g) + H2O(g) (c) C5H6O(I) +...
-
c++. error: array must be initialized with a brace enclosed initializer main.cpp X 10 11 ii 12 13 14 4567 15 16 17 18 19 287288285 20 21 23 24 25 26 27 28 25285922223288 30 31 33 34 36 37 40 41 42 43...
-
C&H Ski Club recently borrowed money and agrees to pay it back with a series of six annual payments of $5,000 each. C&H subsequently borrows more money and agrees to pay it back with a series of four...
-
Which of the following best describes the objectives of Zeros board that the consultant has not yet reviewed? The board should ensure: A. that the assets of the company are used efficiently and...
-
What is a compilation unit?
-
On January 2, 2013, Benson Hospital purchased a $100,000 special radiology scanner from Picard Inc. The scanner had a useful life of 4 years and was estimated to have no disposal value at the end of...
-
Before boarding her flight to Zurich, Switzerland, Mary purchased CHF850 from her bank when the exchange rate was C$1 = CHF0.9651. However, Mary had to cancel the trip. Mary returned to the bank to...
-
Not all potentially good employees have a bubbly, goofy personality of the type that Zappos likes to attract. Would it be wise for the company to reject a candidate solely on the basis of a shy,...
-
Considering your answer to Exercise 79, which type of formula, empirical or molecular, can be obtained from elemental analysis that gives percent composition? Data in Exercise 79, Express the...
-
Express the composition of each of the following compounds as the mass percents of its elements. a. Formaldehyde, CH 2 O b. Glucose, C 6 H 12 O 6 c. Acetic acid, HC 2 H 3 O 2
-
Combine the graphs and equations from questions 3 and 4 and determine the following: a. The equilibrium price of broadband Internet service b. The equilibrium quantity of broadband Internet service...
-
Albee is paid a salary of $33,000 per year and she is paid biweekly. Her regular work week is 35 hours. a) What is Albee's regular hourly rate of pay? b) What is Albee's gross pay for 2 weeks if she...
-
Big Telco Inc. is offering new phones for $200 to customers who also sign up for a three year voice and data plan. The fair value of each phone on a stand-alone basis is $1,000, and each phone costs...
-
Evaluate the following expression using the values mentioned A=0 B=0C = 1 D = 1 F = D(BCA + (AB+C) + C)
-
An Old Office Building in Mandaluyong was purchased by Henry for 120 million pesos. It is that the Gross income of the property is 30 million with a 12% vacancy rate and a credit cost of 1 million....
-
Look at the Microsoft Corporation 2021 proxy statement (for the fiscal year ended June 30, 2021) and the Alphabet Inc. (Google's parent) 2022 proxy statement. Note the differences in styles. For...
-
What is the effect of a stock dividend or a stock split on book value per share?
-
Design and describe an application-level protocol to be used between an automatic teller machine and a bank's centralized computer. Your protocol should allow a user 's card and password to be...
-
An organometallic compound is one containing at least one metalcarbon bond. An example of an organometallic species is (CH 3 CH 2 )MBr, which contains a metalethyl bond. a. If M 2+ has the electron...
-
Esterification reactions are carried out in the presence of a strong acid such as H 2 SO 4 . A carboxylic acid is warmed with an alcohol, and an ester and water are formed. You may have made a...
-
Ignoring ring compounds, which isomer of C 2 H 4 O 2 should boil at the lowest temperature?
-
A phone number in British Columbia consists of one of four area codes (236, 250, 604, and 778), followed by a 7-digit number that cannot begin with a 0 or 1. How many unique phone numbers are there?
-
It takes 5 minutes, 8 seconds to play a song at 120 beats per minute. How long does it take to play the same song at 140 beats per minute? (Round your answer to the nearest second.) 4 minutes 24...
-
This table shows the cost of living in four US cities. One dollar in Detroit has the same buying power as how much money in San Francisco? Round your answer to the nearest cent. Cost of living...

Study smarter with the SolutionInn App


