Calculate the molar mass of the following substances. a. H ON c. (NH4)2Cr2O7 b. HZ
Question:
Calculate the molar mass of the following substances.
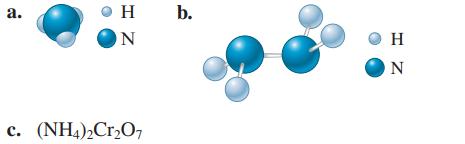
Transcribed Image Text:
a. H ON c. (NH4)2Cr2O7 b. Ο Η HZ N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a NH3 N 14 H 10 1413 17 gmol Molar mass NH...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Calculate the molar mass of the following substances: (a) Li2 CO3, (b) CS2, (c) CHCl3 (chloroform), (d) C6H8O6 (ascorbic acid, or vitamin C), (e) KNO3, (f) Mg3N2.
-
Elaborate figures given below Price per pair (dollars) 105 90 75 60 45 30 15 0 10 20 30 I E Equilibrium IS 40 50 60 70 Quantity of tennis shoes (thousands of pairs per year) 80 90 100 D
-
Richards and Willard determined the molar mass of lithium collected the following data. 6 (a) Find the mean molar mass determined by these workers (b) Find the median molar mass (c) Assuming that the...
-
Fat Gram Calculator Write a flowgorithm program that perform the following tasks: . Ask the user to input the total number of calories in the food the total fat grams in the food Display the...
-
Compute the amount that can be borrowed under each of the following circumstances: 1. A promise to repay $90,000 seven years from now at an interest rate of 6%. 2. An agreement made on February 1,...
-
The objectives of an effective system of corporate governance include all of the following except: A. ensure that the assets of the company are used efficiently and productively. B. eliminate or...
-
Is there a single standard command-line processor to parse and process argv?
-
Lame Specialties manufactures, among other things, woolen blankets for the athletic teams of the two local high schools. The company sews the blankets from fabric and sews on a logo patch purchased...
-
3) (5 marks) Given f(x) = -5x+1 and g(x) = 4x - 3, a. Write an equation for (f + g)(x) b. Determine (f+g)(6) c. Determine (fg)(x) d. Determine (f-g)(x) e. Determine the domain of (fg)(x) 1-x 4) Given...
-
You are the manager of the Mighty Fine mutual fund. The following table reflects the activity of the fund during the last quarter. The fund started the quarter on January 1 with a balance of $40...
-
Aluminum metal is produced by passing an electric current through a solution of aluminum oxide (Al 2 O 3 ) dissolved in molten cryolite (Na 3 AlF 6 ). Calculate the molar masses of Al 2 O 3 and Na 3...
-
A diamond contains 5.0 10 21 atoms of carbon. What amount (moles) of carbon and what mass (grams) of carbon are in this diamond?
-
According to Denison's estimates, which factors were most important in accounting for the growth in real output over the 1929-82 period?
-
What two areas in the Cash Flow report can you determine when a client starts to take supplemental withdrawals from their portfolio?
-
A company that makes soccer balls for professional soccer teams wants to understand the amount of rework in a given manufacturing process. The process has three steps with the following yields: 99%,...
-
Janet's employer provides her a company car. in exchange, janet pays a predetermined amount for personal use of the car. under the commuting value method, what is the least amount that janet must pay?
-
What will be the force of electric repulsion between two small spheres placed 1.0 m apart, if each has a deficit of 108 electrons?
-
Nuclear power plants provide 64.6 % of the total of 5723 MW of electrical energy consumed during a hot summer's day in a certain province. How many megawatts (MW) of electrical energy did the nuclear...
-
Why would a company want to issue callable preferred stock?
-
The baseball player A hits the ball from a height of 3.36 ft with an initial velocity of 34.8 ft/s. 0.14 seconds after the ball is hit, player B who is standing 15 ft away from home plate begins to...
-
In general terms, what does the secondary structure of a protein represent? How is the secondary structure of a protein related to its function?
-
When pure crystalline amino acids are heated, decomposition generally occurs before the solid melts. Account for this observation.
-
When toluene (C 6 H 5 CH 3 ) reacts with chlorine gas in the presence of iron(III) catalyst, the product is a mixture of the ortho and para isomers of C 6 H 4 ClCH 3 . However, when the reaction is...
-
You return from a trip with 190 European euros . How much are your euros worth inU.S.? dollars? Use the exchange rate shownbelow. Currency U.S. dollars per European euro European euros per U.S....
-
A typical day is 24 hours = 1440 minutes = 86,400 seconds. What are the ODDS you will land on 7:15 either AM or PM (twicein a day)? b. What is the PROBABILITY you will land on 7:15 either AM or...
-
In Wisconsin, 30% pass the bar exam on their first try. Of those that take the exam a second time, 40% pass. If you're fresh out of law school, what is the probability of passing the bar within 2...

Study smarter with the SolutionInn App


