Fig. 15.3 outlines the classic scheme for separating a mixture of insoluble chloride salts from one another.
Question:
Fig. 15.3 outlines the classic scheme for separating a mixture of insoluble chloride salts from one another. Explain the chemistry involved in the various steps of the figure.
Fig. 15.3
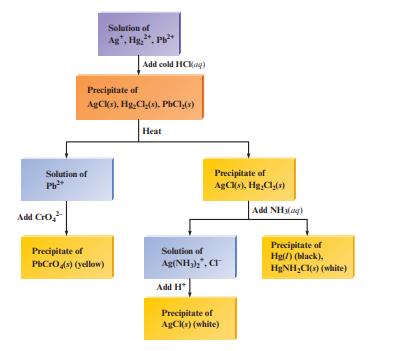
Transcribed Image Text:
Solution of Pb²+ Add CrO₂²- Solution of Ag, Hg., Pb²+ Precipitate of AgCl(s), Hg,Cl(s), PbCl(s) Precipitate of PbCrO4(s) (yellow) Add cold HCKag) Heat Precipitate of AgCl(s), Hg₂Cl(s) Solution of Ag(NH3)2, CT Add H Precipitate of AgCl(s) (white) Add NH3(g) Precipitate of Hg(/) (black). H₂NH₂Cl(s) (white)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The figure you sent me appears to show a separation scheme for a mixture of three ca...View the full answer

Answered By

Rukhsar Ansari
I am professional Chartered accountant and hold Master degree in commerce. Number crunching is my favorite thing. I have teaching experience of various subjects both online and offline. I am online tutor on various online platform.
5.00+
4+ Reviews
17+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Fig. 15.2 summarizes the classic method for separating a mixture of common cations by selective precipitation. Explain the chemistry involved with each of the four steps in the diagram. Fig. 15.2...
-
Outline a procedure for separating a mixture of benzoic acid, 4-methylphenol, aniline,and benzene using acids, bases, and organic solvents.
-
Devise a procedure for separating a mixture of the four stereoisomers of isoleucine into its four components: (+)-isoleucine, (-)-isoleucine, (+)-alloisoleucine, and (-2)-alloisoleucine (Problem 31)....
-
Each of the systems in Problems 11 through 18 has a single critical point (x 0 , y 0 ). Apply Theorem 2 to classify this critical point as to type and stability. Verify your conclusion by using a...
-
If the claimants had refuted the governments assertion that the cash was illicit, would the result in this case have been different? Why or why not?
-
Suppose Intel stock has a beta of 1.6, whereas Boeing stock has a beta of 1. If the risk-free interest rate is 4% and the expected return of the market portfolio is 10%, according to the CAPM, a....
-
Record the following petty cash transactions of Xeno, Corp., in the journal; expla nations are not required. Jun 1 Established a petty cash fund with a $200 balance. 30 The petty cash fund had $22 in...
-
Tiffany Goren started her own consulting firm, Goren Consulting, on May 1, 2010. The trial balance at May 31 is as shown below. In addition to those accounts listed on the trial balance, the chart of...
-
Question 3 A big auto parts distributor imports brake pads from a supplier in Mexico. The monthly demand is stable and is 10,000 units per month. A box costs $20 including shipping but placing an...
-
The graph in Figure 1.38 shows the distance versus time for an elevator as it moves up and down in a building. Compute the elevators velocity at the times marked a, b, and c. 32 28 24 20 16 12 a 28...
-
Calculate the molar solubility of Cd(OH) 2 , K sp = 5.9 10 -11 .
-
Under what circumstances can you compare the relative solubilities of two salts directly by comparing the values of their solubility products? When can relative solubilities not be compared based on...
-
You are one of three carpenters assigned to complete a short construction project. Right before the start of the project, one of your fellow carpenters was hospitalized and will not be available to...
-
Why is propaganda a famous English word? List your answers.
-
How does propaganda pattern help understand the core of the problem?
-
List some of the most notable instances of propaganda in the history of humanity.
-
Is propaganda pattern domain- and application-specific?
-
What is fairness? Can you list different meanings of this word?
-
Required Using Tables I, II, III, or IV in this appendix, calculate the following: a. The future value of $30,000 invested at 8 percent for 10 years. b. The future value of eight annual payments of...
-
Rowland Textile Inc. manufactures two products: sweatshirts and T-shirts. The manufacturing process involves two activities: cutting and sewing. Expected overhead costs and cost drivers are as...
-
Draw a mechanism for the following transformation: NaOH, heat
-
The reaction in the previous problem is an equilibrium process. Draw a mechanism of the reverse process. That is, draw a mechanism showing conversion of the conjugated, cyclic enone into the acyclic...
-
When 2,6-heptanedione is heated in the presence of aqueous sodium hydroxide, a condensation product with a six-membered ring is obtained. Draw the product and show a mechanism for its formation.
-
Gametes 1. The genes for orange and black fur in cats are on the X chromosome. An orange female cat mates with a black male. The resulting litter includes a male calico (orange and black) kitten...
-
Commercial damages, as a result of financial fraud, directly impact the operations of every company. Under this premise: What, in your opinion, could be some of the commercial damages resulting from...
-
Post adjusting entries to the T-accounts. (Post entries in the order of journal entries presented in the previous part.) Cash 7/1 12,700 7/1 2,120 7/21 1,700 7/5 1,920 7/18 1,590 7/20 2,120 7/31 310...

Study smarter with the SolutionInn App


