The Hg 2+ ion forms complex ions with I - as follows: A solution is prepared by
Question:
The Hg2+ ion forms complex ions with I- as follows:
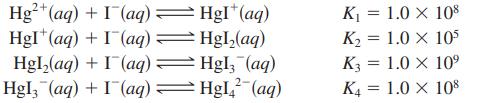
A solution is prepared by dissolving 0.088 mole of Hg(NO3)2 and 5.00 moles of NaI in enough water to make 1.0 L of solution.
![a. Calculate the equilibrium concentration of [HgI4]. b. Calculate the equilibrium concentration of [I]. c.](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1699/9/6/4/82265536796b3c181699964823549.jpg)
Transcribed Image Text:
Hg²+ (aq) + I (aq) = HgI+ (aq) + (aq) = (aq): Hgl₂(aq) + Hgl, (aq) + (aq) → = HgI*(aq) Hgl₂(aq) Hgl, (aq) HgI₂² (aq) K₁ = 1.0 X 108 K₂ = 1.0 X 105 K3 = 1.0 X 109 K4 1.0 X 108
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The Hg2 ion forms complex ions with I as follows Hg2 I HgI HgI I HgI2 HgI2 I HgI3 HgI3 I HgI4 The eq...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
A solution is prepared by dissolving table salt, sodium chloride, in water at room temperature. a. Assuming there is no significant change in the volume of water during the preparation of the...
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
A solution is prepared by dissolving 10.8 g ammonium sulfate in enough water to make 100.0 mL of stock solution. A 10.00-mL sample of this stock solution is added to 50.00 mL of water. Calculate the...
-
Shown here is a list published by Electronics Weekly.com of the top five semiconductor companies in the United States by revenue ($ billions). a. Construct a bar chart to display these data. b....
-
Curtis is an executive on a business trip to the West Coast. He has driven his car on this trip and checks into the Hotel Ritz. The hotel has a guarded underground parking lot. Curtis gives his car...
-
An article in Nuclear Engineering International (February 1988, p. 33) describes several characteristics of fuel rods used in a reactor owned by an electric utility in Norway. Measurements on the...
-
Two conspirators, Timothy Carpenter and Timothy Sanders, were charged with aiding and abetting a robbery affecting interstate commerce and the use or carriage of a firearm. At trial, the FBI...
-
Having rejected a tariff on textiles (a tax on imports), the president of Isoland is now considering the same-sized tax on textile consumption (including both imported and domestically produced...
-
Consider the inverse demand function below for a market with 2 dominant firms: P=600-3(Q 1 +Q 2 ) Assume that both firms have identical marginal costs: $300, yet one of them, Firm 1, is the leader...
-
Cost Estimation at Global Green Books Publishing Global Green Books Publishing is continuing to produce customized eBooks as a key new product line for it as a successful printing and publishing...
-
Calculate the mass of manganese hydroxide present in 1300 mL of a saturated manganese hydroxide solution. For Mn(OH)2, K sp = 2.0 10 -13 .
-
Write equations for the stepwise formation of each of the following complex ions. a. Ni(CN) 4 2- b. V(C2O 4 ) 3 3 -
-
List and describe the courts in the Federal court system and in a typical State court system.
-
A conductor which is 30 cm long with a mass of 20 g is suspended horizontally in a magnetic field whose magnetic flux density is 0.12 T. What current is required in order that the magnetic force...
-
3. A technician is given 120 ml of a 50% (W/V) potassium chloride solution, and is told to add 300 ml of sterile water to it. What will the final (W/V) percentage concentration be of the solution? 4....
-
R Ltd. manufactures three products, A, B and C. The following information is given below. Sales Forecast Product Quantity Price Per Unit A 1000 Rs.100 B 2000 Rs.120 C 1500 Rs.140 Materials Used in...
-
3.1 A pipe with 200mm inner diameter should have a through flow of 10500 standard cubic meters per hour of CO2. ("standard-" or "norm-conditions" are 1.013 bar; 0C). How large are the a) mass flow b)...
-
1. 2. A 40 cm conductor cuts through a magnetic field of 0.50 T at an angle of 60 to the field. If the wire carries a current of 20 A, what is the deflecting force on the conductor? (3.5 N) A single...
-
Carter Hopkins Homes engaged in the following activities during the year: Borrowed $600,000 from Marshall Bank Declared and paid dividends on common stock of $28,000 Made a $78,000 annual...
-
Coastal Refining Company operates a refinery with a distillation capacity of 12,000 barrels per day. As a new member of Coastal's management team, you have been given the task of developing a...
-
Distinguish between the following terms applied to a set of functions: orthogonal and normalized, and orthonormal.
-
Why can we conclude that the wave function (x, t) = (x) e i (E / h )t represents a standing wave?
-
What is the usefulness of a complete set of functions?
-
Why is it that some organizations do not place enough importance on disaster recovery? What are examples of the type of disasters that could happen? What are some things that might happen to these...
-
Leon knows that his first four test grades were 86, 95, 98, and 88. Use the formula x = x1 + x2 + ...+Xn 2 + win to find Leon's grade on the fifth test if his test average is 90.2.
-
How do you know if you would be a good IT project manager? After all, being an IT project manager is a demanding yet rewarding endeavor. Imagine you are chosen to be the IT project manager and must...

Study smarter with the SolutionInn App


