What do each of the following sets of compounds/ions have in common with each other? a. SO3,
Question:
What do each of the following sets of compounds/ions have in common with each other?
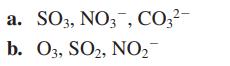
Transcribed Image Text:
a. SO3, NO3, CO3²- b. 03, SO₂, NO₂™
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Lets break it down a SO3 NO3 CO2 All of these compoundsions have one thing in common they are all mo...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
one cannot tell the spatial arrangement because the cells are layered on top of each other
-
What do each of the following sets of compounds/ ions have in common with each other? Reference your Lewis structures for Exercises 88, 91, and 92. a. XeCl4, XeCl2 b. ICl5, TeF4, ICl3, PCl3, SCl2,...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
The temperature of air changes from 0 to 10C while its velocity changes from zero to a final velocity, and its elevation changes from zero to a final elevation. At which values of final air velocity...
-
Web Problem: Go to www.oanda.com/currency/ big-mac-index . Oanda reports the last available Big Mac index but then updates the exchange rates on a regular basis to compare them with the PPP-based...
-
The following data represent the ages of award winners for best actor and best actress in a leading role for the 20 years from 1985 to 2004. (a) Construct an ordered back to back stem-and-leaf...
-
Compare and contrast the ethical approaches of its legal, therefore, its ok and the ends justify the means. Are there similarities? Are there differences?
-
On January 1, 2014, Valdez Company had Accounts Receivable $91,000 and Allowance for Doubtful Accounts $8,100. Valdez Company prepares financial statements annually. During the year, the following...
-
Explain how the relative prices of rugs and robots in autarky compare with the relative prices when Canada and India start to trade? In your answer explain which country will export/import which...
-
The output of the INVERTER of Figure 3-11 is connected to the input of a second INVERTER. Determine the output level of the second INVERTER for each level of input A. Figure 3-11 NOT A 0 1 | x = A 1...
-
Pelargondin is the molecule responsible for the red color of the geranium flower. It also contributes to the color of ripe strawberries and raspberries. The structure of pelargondin is: How many and...
-
(a) Let H G, K G. Show that H V K is normal in G. (b) Prove that the set of all normal subgroups of G forms a complete lattice under inclusion (Introduction, Exercise 7.2). Exercise 7.2 Let G be a...
-
1) If Richard has a $175,000 bond with a 9% interest rate, compounded annually, how much will he have in 7 years?
-
How do intergovernmental and interorganizational systems operate in addressing catastrophic disasters and extreme events?
-
Find the integral to respect x and simplify: S 3x+16x+15 (x+3)3 dx 3+6x+4x-2x (x+3x) x-7x-2 dx x3-8 -dx
-
What are four or five fundamental tenets concerning the influence of student behavior? Explain
-
A stock just paid a dividend of $1.3. The required rate of return is 11.6%, and the constant growth rate is 3%. What is the current stock price? Enter with two decimal places of precision (dollar and...
-
On November 1, Able Corporation purchased a six-month insurance policy from The Baylor Agency for $3,000. a. Prepare the necessary adjusting entry for Able Corporation on November 30, assuming it...
-
What are current assets and current liabilities? How are they different from non-current assets and non-current liabilities?
-
Calculate the pH of a 0.20-M C 2 H 5 NH 2 solution (Kb = 5.6 10 -4 ).
-
Calculate the pH of a 0.050-M (C 2 H 5 ) 2 NH solution (K b = 1.3 10 -3 ).
-
What is the percent ionization in each of the following solutions? a. 0.10 M NH 3 b. 0.010 M NH 3 c. 0.10 M CH 3 NH 2
-
The present value of an investment is estimated at about $ 2 6 6 , 3 0 0 . The expected generated free cash flow from the project for next year is $ 5 , 0 0 0 and is expected to grow 1 5 % a year for...
-
A 6.0-kg block situated on a frictionless incline is connected to a light spring (k = 200 N/m), as shown. The block is released from rest when the spring is unstretched and slides down 0.3 m. The...
-
A crate with a mass of M = 85.5 kg is suspended by a rope from the midpoint of a uniform boom. The boom has a mass of m = 122 kg and a length of I= 7.95 m. The end of the boom is supported by another...

Study smarter with the SolutionInn App


