Write the symbol of each atom using the format. A ZA
Question:
Write the symbol of each atom using the ![]() format.
format.
Transcribed Image Text:
A ZA
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (3 reviews)
a 23 Na 10 electrons 10 p...View the full answer

Answered By

Leah Muchiri
I am graduate in Bachelor of Actuarial Science and a certified accountant. I am also a prolific writer with six years experience in academic writing. My working principle are being timely and delivering 100% plagiarized free work. I usually present a precised solution to every work am assigned to do. Most of my student earn A++ GRADE using my precised and correct solutions.
4.90+
52+ Reviews
125+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Write a policy statement as the HR director stating whether or not office romantic relationships are allowed. If so, under what circumstances? What theoretical ethical perspective did you use to...
-
This exercise is comprised of four parts. Part 1 covers automated routines macros in Excel to rearrange and transform data, Part 2 covers Pivot Table: Data Sets Cross-Tabulation and Querying in Excel...
-
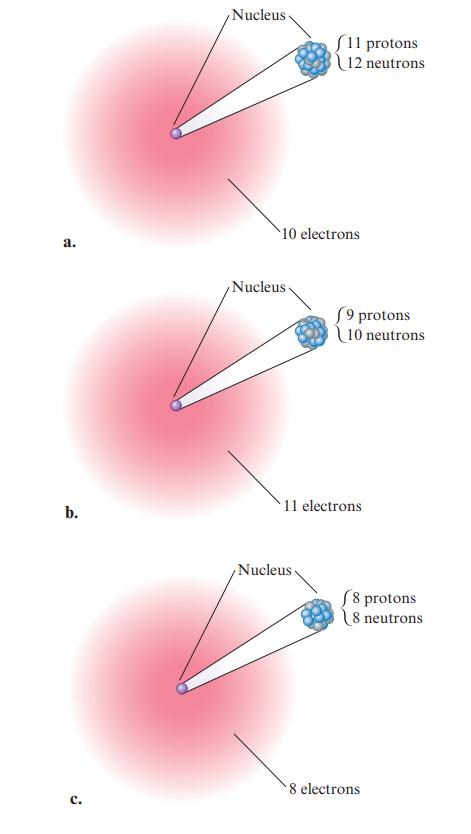
Consider the following depictions of two atoms, which have been greatly enlarged so you can see the subatomic particles. a. How many protons are present in atom A? b. What is the significance of the...
-
Daniel and Karen Chapman have three children, aged 2, 8 and 11 at the end of the year. The 8 year old is blind and therefore qualifies for the disability tax credit. The other two are in good mental...
-
Would Oregon Co.s real cost of hedging Australian dollar payables every 90 days have been positive, negative, or about zero on average over a period in which the dollar weakened consistently? What...
-
Novo Nordisk is a Denmark-based biopharmaceutical company with a focus on diabetes drugs. The company provides detailed disclosure of revenue along geographic, business segment, and product lines....
-
You need to understand the approach described in question 3 in More Genetic TIPS before answering this question. A gene that is normally expressed in pancreatic cells was cloned and then subjected to...
-
The unadjusted trial balance of Aurora Air Purification System at December 31, 2016, and the data needed for the adjustments follow. Adjustment data at December 31 follow: a. On December 15, Aurora...
-
(a) Let3+ and 2 = a + bi be complex numbers. Suppose that 7 Argument = 12' find Argument(22). 7-2 (b) Let the map f: CC be defined by f(z) = Find f() if=1+2i. 1 (c) Solve the equation -12 i(9-2),...
-
What is the modern view of the structure of the atom?
-
Explain the law of conservation of mass and the law of definite proportion.
-
Write an equation for the expected products of 1,2-addition and 1,4-addition of bromine, Br2, to 1,3-butadiene.
-
6.2 Generation of short laser pulses by Quality factor (Q) switching. (a) What is the energy Ep and power Pp of a Q-switched laser pulse (hint: use energy balance with An the inversion built up until...
-
Dr. Erickson's office has benefited from using a clearinghouse to submit claims for payment. They are receiving payments quicker now. Another benefit is: a. patients can submit their own claims. O b....
-
Consider the vector =-5.89+-6.16+-0.26. What is ( ) ? X -0.07 Correct Answer: 0.27
-
Provide a fully worked out numerical example where you characterize the optimal investment and the optimal consumption decisions of a rational agent, who has a given availability of resources over...
-
6. What advantages does a pivoting axle suspension system entail? 7. Could you class pneumatic tyres as a form of suspension. Explain your answer True/False 8. What special precaution must be...
-
For each of the following independent Cases A and B, fill in the missing information. The company budgets and applies production-overhead costs on the basis of direct-labor hours. (U denotes...
-
Prove that the mean heat capacities C P H and C P S are inherently positive, whether T > T 0 or T < T 0 . Explain why they are well defined for T = T 0 .
-
Show how Boyles law, Charless law, and Avogadros law are special cases of the ideal gas law. Using the ideal gas law, determine the relationship between P and n (at constant V and T) and between P...
-
Explain how a barometer and a manometer work to measure the pressure of the atmosphere or the pressure of a gas in a container.
-
Write the balanced formula equation for the acidbase reactions that occur when the following are mixed. a. Potassium hydroxide (aqueous) and nitric acid b. Barium hydroxide (aqueous) and hydrochloric...
-
=180 7. A uniform drawbridge is held by a strong cable in static equilibrium as shown. The length cal the drawbridge is L = 20 m and its mass is m = 200 kg. The angle it makes with the vertical wall...
-
A basketball player jumps straight up for a ball. To do this, he lowers his body 0.330 m and then accelerates through this distance by forcefully straightening his legs. This player leaves the floor...
-
What practical problems are presented by the First Amendment's freedom of the press? What does the term religion mean under the First Amendment? Could you use Constitutional Law: Govermental Powers...

Study smarter with the SolutionInn App


