A 0.20 mol sample of magnesium burns in air to form 0.20 mol of solid MgO. What
Question:
A 0.20 mol sample of magnesium burns in air to form 0.20 mol of solid MgO. What amount (moles) of oxygen (O2) is required for a complete reaction?
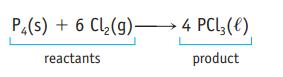
Transcribed Image Text:
P4(s) + 6 Cl₂(g) 4 PCL3 (1) reactants product
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
In the chemical reaction between magnesium Mg and o...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Magnesium burns in air to produce magnesium oxide, MgO, and magnesium nitride, Mg3N2. Magnesium nitride reacts with water to give ammonia. Mg3N2(s) + 6H2O(l ) 3Mg(OH)2(s) + 2NH3(g) What volume of...
-
A 21.496-g sample of magnesium is burned in air to form magnesium oxide and magnesium nitride. When the products are treated with water, 2.813 g of gaseous ammonia are generated. Calculate the...
-
(a) Define the following in terms of gain or loss of hydrogen with one example each : (i) oxidation (ii) reduction (b) When a magnesium ribbon is heated, it burns in air to form magnesium oxide....
-
Question 2A) Explain the differences between right issues and bonusissues. Your answer should include the advantages and disadvantagesof both right issues and bonus issues. [ 8marks}b) Explain pos 0...
-
Suppose that upon Polands entering the European Union, it is discovered that the cost of automobile production in Poland is 20,000 while it is 30,000 in Germany. Suppose that the EU, which has a...
-
1. What important internal controls were ignored when LJM1 was created? 2. How might Enron's harsh Performance Review Committee have aided company executives in committing the fraud? 3. The fraud at...
-
Verify the entries in Table 13.8 for the gamma distribution. Specifically: a. Show that the gamma is a member of the linear exponential family of distributions. b. Describe the components of the...
-
Explain why a survey of the current system may no purpose when an organization is planning to implement an ERP.
-
One possible waste product of a nuclear reactor is the nuclide caesium-137 which decays to form a nuclide of barium (Ba). a) State the nuclear equation for this decay. b) State three quantities that...
-
Equal amounts of two acidsHCl and HCO 2 H (formic acid)are placed in aqueous solution. When equilibrium has been achieved, the HCl solution has a much greater electrical conductivity than the HCO 2 H...
-
Oxidation of 1.00 g of carbon monoxide, CO, produces 1.57 g of carbon dioxide, CO 2 . How many grams of oxygen were required in this reaction? P4(s) + 6 Cl(g) 4 PCL3 (1) reactants product
-
Lloyd owns a beach house (four years) and a cabin in the mountains (six years). His adjusted basis is $300,000 in the beach house and $315,000 in the mountain cabin. Lloyd also rents a townhouse in...
-
What will be the moment of inertia of a homogeneous triangular board of mass m with side lengths a and b when rotated about the axis passing through side a? mb mb mb mb (a) (b) 2mb 3 (c) (e) 3 12 2 6...
-
10. A ray of light falls on a horizontal plane mirror as shown. 40 In order to reflect the light ray vertically upwards, the plane mirror should be rotated by A. 20 B. 25 C. 40 D. 50
-
You are at a funhouse at Hershey Park. The ride you are on is a huge spinning platform.Suppose you move towards the outside edge of the spinning platform. What would happen to the angular momentum of...
-
A tennis ball is tossed straight up into the air. In addition to the force of gravity, the tennis ball is subject to a frictional force due to air friction. The time the tennis ball takes to reach...
-
The capacitor in the circuit is charged to 20 V at the time the switch is closed. (Figure 1) Figure 20 V C 1 0 12 x 10 i 20 1 of 1 80 www Part A If the capacitor ruptures when its terminal voltage...
-
A survey conducted by the Ethics Resource Center discovered that 35% of all workers say that coworkers have committed some kind of office theft. Suppose a survey is conducted in your large company to...
-
Pearson Education, a publisher of college textbooks, would like to know if students prefer traditional textbooks or digital textbooks. A random sample of students was asked their preference and the...
-
One mole of an ideal gas is subjected to the following changes. Calculate the change in temperature for each case if C V,m = 3/2R. a. q = 425 J, w = 185 J b. q = 315. J, w = 315 J c. q = 0, w = 225 J
-
Consider the adiabatic expansion of 0.500 mol of an ideal monatomic gas with C V ,m = 3/2R. The initial state is described by P = 6.25 bar and T = 300.K. a. Calculate the final temperature if the gas...
-
A pellet of Zn of mass 31.2 g is dropped into a flask containing dilute H 2 SO 4 at a pressure of P = 1.00 bar and temperature of T = 300.K. What is the reaction that occurs? Calculate w for the...
-
How can organizational leaders foster a culture that encourages and sustains advanced OCB among employees?
-
The state is beginning a new apprenticeship program for incarcerated individuals. Officials designed the program to improve the chances of inmates finding a job upon release and to increase the wages...
-
If the inflation Earnings on index at the first quarter (before resetting) was 120, what would the equivalent 2nd quarter figure be?

Study smarter with the SolutionInn App


