A gas chromatograph is calibrated for the analysis of isooctane (a major gasoline component) using the following
Question:
A gas chromatograph is calibrated for the analysis of isooctane (a major gasoline component) using the following data: 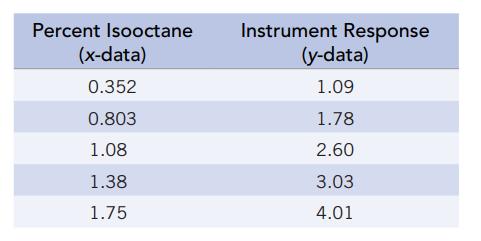
If the instrument response is 2.75, what percentage of isooctane is present?
Transcribed Image Text:
Percent Isooctane (x-data) 0.352 0.803 1.08 1.38 1.75 Instrument Response (y-data) 1.09 1.78 2.60 3.03 4.01
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
To determine the percentage of isooctane x corresponding to an instrument response of 275 y we can u...View the full answer

Answered By

Lilian Nyambura
Hi, am Lilian Nyambura, With extensive experience in the writing industry, I am the best fit for your writing projects. I am currently pursuing a B.A. in Business Administration. With over 5 years of experience, I can comfortably say I am good in article writing, editing and proofreading, academic writing, resumes and cover letters. I have good command over English grammar, English Basic Skills, English Spelling, English Vocabulary, U.S. English Sentence Structure, U.K. or U.S. English Punctuation and other grammar related topics. Let me help you with all your essays, assignments, projects, dissertations, online exams and other related tasks. Quality is my goal.
4.80+
378+ Reviews
750+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
A gas chromatograph is an instrument that measures the amounts of various compounds contained in a sample by separating the various constituents. Because different components are flushed through the...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Federal government budgeting is the forecast or estimate of revenue and expenditures for each fiscal year. The federal budget, like a household budget, itemizes government spending for the following...
-
How does the range of hFE (Fig. 3.23j, normalized from hFE = 100) compare with the range of hfe (Fig. 3.23f) for the range of IC from 0.1 to 10 mA?
-
Anchor.com, a computer consulting firm, has decided to write off the $7,130 balance of an account owed by a customer. Journalize the entry to record the write-off, (a) Assuming that the allowance...
-
Arabica Highland Coffee Company roasts and packs coffee beans. The process begins by placing coffee beans into the Roasting Department. From the Roasting Department, coffee beans are then transferred...
-
Fine Candles Ltd has prepared the following draft profit analysis for the current year. Required Answer each of the following four independent situations. (a) If the companys manager is considering...
-
When Orlandos Arnold Palmer Hospital began plans to create a new 273-bed, 11-story hospital across the street from its existing facility, which was bursting at the seams in terms of capacity, a...
-
x- y 5. (5) Let f(x, y) = a. Sketch the level curve of f where z = 0. b. Use one path to come up with a conjecture for the following limit and then use the epsilon-delta definition of a limit to...
-
A general chemistry class carried out an experiment to determine the percentage (by mass) of acetic acid in vinegar. Ten students reported the following values: 5.22%, 5.28%, 5.22%, 5.30%, 5.19%,...
-
You can analyze for a copper compound in water using an instrument called a spectrophotometer. [A spectrophotometer is a scientific instrument that measures the amount of light (of a given...
-
In a recent year, Tapestry Inc., parent company of major fashion brands Coach New York, Kate Spade New York, and Stuart Weitzman, issued 1,300,000 shares of its $0.01 par value stock for $10,500,000...
-
Risk-free zero-coupon government bonds have the following terms and yields to maturity Term to Maturity 1 year 2 years 3 years 4 years 5 years Yield to Maturity 3.0% 4.0% 5.0% 4.5% 4.0% (a.) Find the...
-
You buy a nine-year bond for $940. The bond has a 3.10% coupon rate. Coupons are s/a. What is the bond's yield to maturity?
-
Charges q 1 , q 2 , and q 3 are all along the same line with q 1 on the left, q 3 on the right, and q 2 exactly midway between q 1 and q 3 . q 1 = 1 . 0 C , q 2 = 2 . 0 C and q 3 = + 4 . 0 C . What...
-
Two dimensions. In the figure, three point particles are fixed in place in an xy plane. Particle A has mass mA = 4 g, particle B has mass 2.00mA, and particle C has mass 3.00mA. A fourth particle D,...
-
Exhibit below for Frits Seegers Inc. and measure the free cash flow for Seegers for Year 0 and six years of forecasts (Year +1 to Year +6) shown. The company does not hold any excess cash, so the...
-
Nickleson Company had an unadjusted cash balance of $7,750 as of May 31. The companys bank statement, also dated May 31, included a $72 NSF check written by one of Nicklesons customers. There were...
-
In a paragraph of approximately 150-200 words, analyze a film or TV/Streaming Show poster of your choosing by focusing on the ways in which representations in the poster are gendered. Include an...
-
Identify the reagents that you would use to accomplish each of the following transformations: a. b. c. d. e. f. -Br OH
-
When the following ester is treated with lithium iodide in DMF, a carboxylate ion is obtained: (a) Draw the mechanism of this reaction. (b) When the methyl ester is used as the substrate, the...
-
Suggest an efficient synthesis for the following transformation: En Br
-
Police Corp. Station Corp. Consolidation Entries DR CR Consolidated Income Statement Sales 200,000 Less: Depreciation Expense (25,000) Less: Other Expenses (105,000) Income from Station Corp. 30,000...
-
Compound interest (Unit4B) produces exponential growth because an inte rest-bearing account grows by the same percentage each year. Suppose your bank account has a doubling time of 21 years. By what...
-
A loan was made in 40 months at 6% simple interest. The principal of the loan has just been repaid along with 800 of interest. Compute the principal amount of the loan

Study smarter with the SolutionInn App


