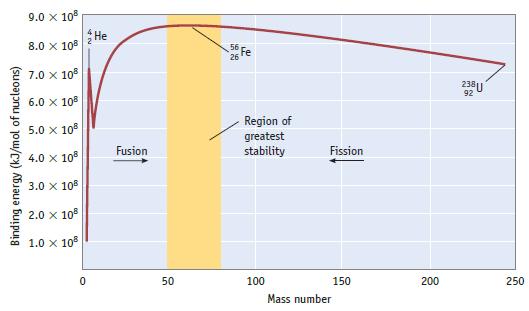
A graph of binding energy per nucleon is shown in Figure 25.4. Explain how the data used
Question:
A graph of binding energy per nucleon is shown in Figure 25.4. Explain how the data used to construct this graph were obtained.
Data given in Figure 25.4
Transcribed Image Text:
Binding energy (kJ/mol of nucleons) 9.0 x 108 8.0 x 108 7.0 x 108 6.0 x 108 5.0 X 108 4.0 x 108 3.0 X 108 2.0 x 108 1.0 x 108 0 He Fusion 50 56 Fe Region of greatest stability 100 Fission 150 Mass number 200 238U 92 250
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
The data used to construct the graph of binding energy per nucleon in Figure 254 were obtained from a series of experiments that were conducted at the ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Consider the following graph of binding energy per nucleon as a function of mass number. a. What does this graph tell us about the relative halflives of the nuclides? Explain your answer. b. Which...
-
The most stable nucleus in terms of binding energy per nucleon is 56Fe. If the atomic mass of 56Fe is 55.9349 amu, calculate the bind mg energy per nucleon for 56Fe.
-
The isotope has the largest binding energy per nucleon of any isotope. Calculate this value from the atomic mass of nickel-62 (61.928345 amu) and compare it with the value given for iron-56 in Table...
-
The Black Sheep Inc.'s call option has the following characteristics: Exercise price of The Black Sheep Inc.'s call option ($) 75 Annualized risk-free rate 4.5% Time to the expiration of the options...
-
Titanium Metals Inc. casts blades for turbine engines. Within the Casting Department, alloy is first melted in a crucible, then poured into molds to produce the castings. On October 1, there were 700...
-
Xeroxs bundled sales were improperly recorded in its financial statements because: (a) All of the revenue streams for its leases should have been recorded over the life of the leases. (b) Some of the...
-
Krupp Refrigeration, Inc., is trying to reduce inventory and wants you to install a kanban system for compressors on one of its assembly lines. Determine the size of the kanban and the number of...
-
Graves Mining Company declared, on April 20, a dividend of $500,000 payable on June 1. Of this amount, $125,000 is a return of capital. Prepare the April 20 and June 1 entries for Graves.
-
You are driving your car at a speed of 35.0 m/s and you hit the brakes slowing to 20.0 m/s. If it takes 4.50 s to slow down (a) Determine the acceleration of the car. (b) Determine the displacement...
-
What information was used to identify and particles?
-
Rank the three types of natural radiation (, , ): (a) In order of increasing mass (b) In order of increasing penetrating power
-
Which of these events is an auditor not required to communicate to a companys audit committee or board of directors? a. Managements significant accounting policies. b. Managements judgments about...
-
Explain how group criminality differs from complicity and vicarious liability.
-
Which should we be more concerned with: traditional property damage and invasion, or property damage and invasion with computers?
-
Why do some states treat armed robbery different than other forms of robbery?
-
In April 2002, Stallings was incarcerated at the Algoa Correctional Center, having served thirteen years of his twenty-three year sentence for various felony offenses. He worked as a clerk in the...
-
What is the difference between physician-assisted suicide and euthanasia? Which, if either, is criminal?
-
Davis Co. uses backflush costing to account for its manufacturing costs. The trigger points are the purchase of materials, the completion of goods, and the sale of goods. Prepare journal entries to...
-
Which one of the following anhydrous chloride is not obtained on direct heating of its hydrated chloride? (A) BaCl2 (B) CaClz (C) MgCl2 (D) SrCl2
-
Find i and V o in the circuit of Fig. 2.100 . 80 2 24 Q ww 25 2 30 : 20 20 V V. 20 2 60
-
Calculate V o and I o in the circuit of Fig. 2.99. 30 70 200 V 20 5
-
Using series/parallel resistance combination, find the equivalent resistance seen by the source in the circuit of Fig. 2.98 . Find the overall absorbed power by the resistor network. 70 2 50 2 150 2...
-
Salvia Company recently purchased a truck. The price negotiated with the dealer was $40,000. Salvia also paid sales tax of $2,000 on the purchase, shipping and preparation costs of $3,000, and...
-
Hazelnut Corporation manufactures lawn ornaments. It currently has two product lines, the basic and the luxury. Hazelnut has a total of $143,835 in overhead. The company has identified the following...
-
White Corporation's budget calls for the following sales for next year: Quarter 1 Quarter 2 105,500 units 88,000 units Quarter 3 Quarter 4 71,900 units 103,600 units Each unit of the product requires...

Study smarter with the SolutionInn App


