Balance each of the following unbalanced equations; then calculate the standard potential, E, and decide whether each
Question:
Balance each of the following unbalanced equations; then calculate the standard potential, E°, and decide whether each is product-favored at equilibrium as written. (All reactions are carried out in acid solution.)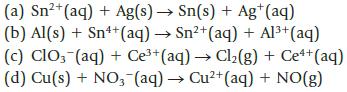
Transcribed Image Text:
2+ (a) Sn²+ (aq) + Ag(s) → Sn(s) + Ag+ (aq) (b) Al(s) + Sn++ (aq) → Sn²+ (aq) + Al³+ (aq) (c) ClO3(aq) + Ce³+ (aq) → Cl₂(g) + Ce++ (aq) (d) Cu(s) + NO3(aq) → Cu²+ (aq) + NO(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To balance these unbalanced redox equations and determine whether they are productfavored at equilib...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Balance each of the following unbalanced equations; then calculate the standard potential, E, and decide whether each is product-favored at equilibrium as written. (All reactions are carried out in...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
A pack of iron bolts is such that the difference in masses or weights of successive sizes is the same bolt being of mass 13.5 grams and the largest is 94.5 grams. If the total mass of the complete...
-
H.J. Heinz Company uses standards to control its materials costs. Assume that a batch of ketchup (1,500 pounds) has the following standards: The actual materials in a batch may vary from the standard...
-
Consider a risk-neutral firm, protected by limited liability, that wants to finance a project at a cost \(I=1\). The project takes one period to complete. The firm has no initial wealth; hence to...
-
Explain the nature of the first six types of Braitenberg vehicles. Discuss how these vehicles can help us to understand the nature of intelligence.
-
Suppose that technophiles are willing to pay $400 now for the latest iPhone, but only $300 if they have to wait a year. Normal people are willing to pay $250, and their desire to purchase does not...
-
One of the products that the sporting - goods retail chain, Sports World, sells is parachutes. In the first quarter of 2 0 2 4 , Sports World sold 6 5 0 parachutes. Budgeted sales for the remainder...
-
Consider the following half-reactions: (a) Based on E values, which metal is the most easily oxidized? (b) Which metals on this list are capable of reducing Fe 2+ (aq) to Fe(s)? (c) Write a balanced...
-
Calculate the value of E for each of the following reactions. Decide whether each is product-favored at equilibrium in the direction written. [Reaction (d) is carried out in basic solution.] (a)...
-
A dynamic model of a continuous-flow, biological chemostat has the form where X is the biomass concentration, S is the substrate concentration, and C is a metabolic intermediate concentration. The...
-
Dale, an assistant scoutmaster for the Boy Scouts, lost his position when the organization learned that he was gay. Dale contended that he was the victim of discrimination while the Boy Scouts argued...
-
A business in which there are one or more general partners and one or more limited partners. a. articles of incorporation b. corporation c. duty of care d. duty of loyalty e. partnership f. joint v...
-
In a well-known paper, Roll (1978) discusses tests of the SML in a four-asset context: a. Derive two efficient portfolios in this 4-asset model and draw a graph of the efficient frontier. b. Show...
-
Figure 2.5 illustrates the interface lines of a generic memory component. Assume m = 15 and n = 7. The address bus of your microprocessor is 24 bits wide. How, in principle, could you locate this...
-
Show with an illustrative example how the five - stage pipeline discussed in this chapter (Fig. 2.10 ) could benefit from the Harvard architecture. Figure 2.10 FDLE S FDLES FDLES F DLES FDLES FDLES...
-
Forming, the second department in a three-department production process for Chula Vista Can Inc., received 15,000 units with a total cost of $45,000 from Blanking during the month of May. Production...
-
Smthe Co. makes furniture. The following data are taken from its production plans for the year. Required: 1. Determine the hazardous waste disposal cost per unit for chairs and for tables if costs...
-
Slap shot. In professional hockey, a slap shot is a type of shot in which a player literally slaps the puck with his stick. The puck may start with a very low speed and can leave the stick with a...
-
A spring scale indicates that a helium balloon tied to it produces a tension of 0.20 N in the string. The string is then cut, and the balloon rises until it comes to rest on the ceiling. (a) Draw a...
-
A subway train is designed with a maximum acceleration of +0.20 m/s 2 , which allows for both passenger safety and comfort. (a) If subway stations are 1.2 km apart, what is the maximum velocity that...
-
The most recent financial statements for Martin, Inc., are shown here: Income Statement Sales Costs Taxable income Taxes (21%) Net income $24,550 -14,730 $9,820 -2,062 $7,758 Assets Balance Sheet...
-
Draw a finite automata over alphabet {a, b} for { w | every letter in w is either preceeded or followed by the sameletter} For example aab and aabaa are not in the set because the b isneither...
-
Kapono Farms exchanged an old tractor for a newer model. The old tractor had a book value of $15,000 (original cost of $34,000 less accumulated depreciation of $19,000) and a fair value of $9,600....

Study smarter with the SolutionInn App


