Balance the following equations, and then write the net ionic equation. (a) (NH4)2CO3(aq) + Cu(NO3)2(aq) CuCO3(s)
Question:
Balance the following equations, and then write the net ionic equation. 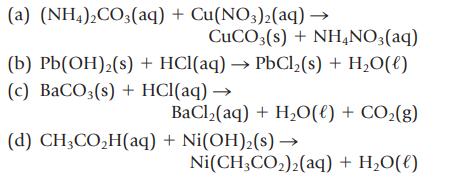
Transcribed Image Text:
(a) (NH4)2CO3(aq) + Cu(NO3)2(aq) → CuCO3(s) + NH4NO3(aq) (b) Pb(OH)₂(s) + HCl(aq) → PbCl₂(s) + H₂O(l) (c) BaCO3(s) + HCl(aq) → BaCl,(aq) + H,O(l) + CO,(g) (d) CH3CO₂H(aq) + Ni(OH)₂(s) → Ni(CH3CO₂)2(aq) + H₂O(l)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a NH42CO3aq CuNO32aq CuCO3s NH4NO3aq Balanced equation NH42CO3aq CuNO32aq CuCO3s 2NH4NO3aq Net ionic ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) CH3COOH (aq) + KOH(aq)- .co.(aq) + NaO H (aq) - HNO3(aq ) + Ba(OH)2(aq )-
-
1) Write balanced net ionic equation for the neutralization ofequal molar amounts of HNO2 and KOH 2) Write balanced net ionic equation for the neutralization ofequal molar amounts of HBr and NH3 3)...
-
In the dynamic and expanding urban environment of "Slothsberg", a new highway ("Snailpace Highway") is to be constructed over the existing "Dillydally Highway". The following conditions apply to the...
-
Return to the example in this chapters final Case Study of how a 10 percent dollar depreciation affects U.S. net foreign wealth (page 316). Show the size of the effect on foreigners net foreign...
-
A tidal wave or tsunami is usually the result of an earthquake in the Pacific Rim, often 1000 or more miles from Hawaii. Tsunamis are rare but dangerous. Many tsunamis are small and do little damage....
-
The ratio of indicated thermal efficiency to the corresponding air standard cycle efficiency is called (a) net efficiency (b) efficiency ratio (c) relative efficiency (d) overall efficiency
-
The director of project management calls you into his office and informs you that one of your fellow project managers has had a severe heart attack midway through a project. You will be taking over...
-
Gerald Sales has total owner's equity of $18,700. The firm has current liabilities of $2,500 and total assets of $31,800. What is the value of the long-term debt, if you also know that market value...
-
Write an equation that describes the equilibrium that exists when the weak acid benzoic acid (C 6 H 5 CO 2 H) dissolves in water. Identify each of the four species in solution as either Brnsted acids...
-
Write an equation that describes the equilibrium that exists when nitric acid dissolves in water. Identify each of the four species in solution as either Brnsted acids or Brnsted bases. Does the...
-
Examine the data encryption technique illustrated in Figure 14-10. Use a displacement value of ?8? to encrypt the following message. Hint: This task becomes easy if you use an Excel spreadsheet and...
-
explain why we would want the exact solution to a real-world or mathematical problem. explain why we would want an approximate solution to a real-world or mathematical problem?
-
You have a portfolio consisting solely of shares G and share W. The portfolio has an expected return of 13.64%. Share G has an expected return of 8.2% while share W is expected to return 16.2%. What...
-
Johnson was known for "having trouble remembering what was his and what was the company's." For example, the company had a fleet of 10 corporate planes (also called the RJR air force), used for...
-
Jargon Jen has five million shares outstanding, generates free cash flows of $12 million each year and has a cost of capital of 6%. It also has $3 million of cash on hand. Jargon Jen wants to decide...
-
Metabolizing a candy bar releases about 1 0 ^ 6 joules of energy. Assume the candy bar's mass is 0 . 3 6 kg and use the formula kinetic energy = 1 / 2 mv ^ 2 , where m is the object's mass and v is...
-
Refer to the preceding information for Paulcrafts acquisition of Switzers common stock. Assume that Paulcraft pays $420,000 for 70% of Switzer common stock. Paulcraft uses the cost method to account...
-
Explain why each of the following is either a private good or a public good: traffic lights, in line skates, a city park, a chicken salad sandwich, a tennis racket, national defense, a coastal...
-
Identify the reactants that you would use to make each of the following enamines: (a) (b) (c) -N
-
Predict the product of the two-step procedure below, and draw a mechanism for its formation: 1) [H1, -N-NH2 -H2 2) / -0, heat
-
Propose a plausible mechanism for each of the following hydrolysis reactions: (a) (b) (c) (d) EtO OEt * * + 2 ELOH (b) N. * .N'
-
Encouraging team productivity can be a major issue in many organizations. Inclusive and Exclusive are the two common management styles most organizations exhibit. Discuss the effectiveness or...
-
Larry Culp Redefines Success at GE When Larry Culp became the 12th chief executive officer of General Electric, he was the first CEO to be brought in from outside the company, and GE desperately...
-
You have been meeting on a team for the past couple of weeks, and while you really get along well with your teammates, it is clear that very little "work" is getting done at these meetings. The other...

Study smarter with the SolutionInn App


