Balance the following equations involving organic compounds. (a) Ag*(aq) + CH;CHO(aq) Ag(s) + C6H5COH(aq) (b) CHCHOH +
Question:
Balance the following equations involving organic compounds.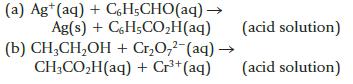
Transcribed Image Text:
(a) Ag*(aq) + CH;CHO(aq)— Ag(s) + C6H5CO₂H(aq) (b) CH₂CH₂OH + Cr₂O,2 (aq) → CH3CO₂H(aq) + Cr³+ (aq) (acid solution) (acid solution) 1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Balanced equations a Ag aq C6H5CHOaq AgC6H5O2s CH3CO2Haq acid solution b CH3CH2OH Cr2O72aq CH2CO2Haq ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Pecunious Products, Inc.'s financial results for the past three years are summarized below: Year 3 Year 2 Year 1 Sales trend 179.2 161 140 Current ratio 3.5 3.22 3.08 Acid-test ratio 1.12 1.26 1.54...
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) CH3COOH (aq) + KOH(aq)- .co.(aq) + NaO H (aq) - HNO3(aq ) + Ba(OH)2(aq )-
-
In the diagram, the positive terminal of the 12 V battery is grounded it is at zero potential. At what potential is point X? 12 V 4 V Ground
-
Twelve states, some local governments, and private organizations sued the EPA, contending that it did not live up to its obligation under the Clean Air Act to regulate greenhouse gases that result...
-
Independence Corporation needs to replace some of the assets used in its trade or business and is contemplating the following exchanges: Determine whether each exchange qualifies as a like-kind...
-
True or False. Gathering data is all that is required for a successful trending program.
-
Preparing a statement of cash flows. (Adapted from CPA examination.) The management of Warren Corporation, concerned over a decrease in cash, provides you with the comparative analysis of changes in...
-
Over the course of the semester, we covered a clear and effective 10 Step process for developing a strategy as described in the Inspired. Logical. Strategy That Works book. Please bullet point the 10...
-
A voltaic cell is constructed in which one half-cell consists of a silver wire in an aqueous solution of AgNO 3 . The other half-cell consists of an inert platinum wire in an aqueous solution...
-
Write balanced equations for the following reduction half-reactions involving organic compounds. (a) HCOH CHO (b) C6H5COH C6HCH3 (c) CH,CH,CHO (d) CH3OH CH CHCHCHOH (acid solution) (acid solution)...
-
In Exercises 35 through 44, use calculus to sketch the graph of the given function. g(t) = 1 + 3
-
The IRC allows individuals and businesses to make noncash contributions to qualifying charities and to claim deductions for these contributions on their income tax returns. The Internal Revenue...
-
Investment portfolio rebalancing is part of which phase in the investment planning process? I. Planning. II. Implementation. III. Monitoring. IV. Updating. A. I. B. I, II. C. III. D. II, IV.
-
Robert purchased a single life annuity for \($100,000.\) His lifetime monthly payment is \($1,000.\) According to actuarial tables, Roberts life expectancy is 10 years. Which is correct? A. If Robert...
-
Investors in real estate seek which objective? A. High correlation with common stock. B. Liquidity. C. Long-term growth. D. Low correlation with inflation.
-
Regarding contributions to an HSA, which of the following statements is incorrect? A. Contributions can be made by the employer or the individual, or both. B. Contributions made by the employer are...
-
United Technologies Corporation provides a broad range of high-technology products and support services to the building systems and aerospace industries. Those products include Pratt & Whitney...
-
Maria Castigliani is head of the purchasing department of Ambrosiana Merceti, a medium-sized construction company. One morning she walked into the office and said, The main problem in this office is...
-
Quasimonochromatic light having an irradiance of 400 W/m 2 is incident normally on the cornea (n c = 1.376) of the human eye. If the person is swimming under the water (n w = 1.33), determine the...
-
Compare the amplitude reflection coefficients for an airwater (n w = 4/3) interface with that of an aircrownglass (n g = 3/2) interface, both at near-normal incidence. What are the corresponding...
-
Use Eq. (4.42) and the power series expansion of the sine function to establish that at near-normal incidence we can obtain a better approximation than the one in Problem 4.45, which is [-r ¥ ]...
-
Jason is completing a feasibility study for a new restaurant. He estimates that the restaurant will achieve an average spend per cover of $32.50 and the Cost of Goods Sold will be $11.20. If the...
-
How do social networks contribute to socioeconomic stratification and echo chambers, and what are the broader societal implications ?
-
Find f'(x) and f'(c). Function f(x) = (x5+ 5x)(2x5 + 2x - 4) Value of c C = 0 f'(x) = f'(c) =

Study smarter with the SolutionInn App


