Calculate r G and the equilibrium constant for the following reactions. (a) 2 Fe+ (aq) +
Question:
Calculate ΔrG° and the equilibrium constant for the following reactions.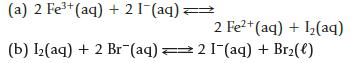
Transcribed Image Text:
(a) 2 Fe³+ (aq) + 21 (aq): (b) 1₂(aq) + 2 Br(aq) = 2 Fe²+ (aq) + ₂(aq) 21-(aq) + Br₂(l)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To calculate rG standard Gibbs free energy change and the equilibrium constant K for these reactions ...View the full answer

Answered By

Anthony Ngatia
I have three academic degrees i.e bachelors degree in Education(English & Literature),bachelors degree in business administration(entrepreneurship option),and masters degree in business administration(strategic management) in addition to a diploma in business management.I have spent much of my life in the academia where I have taught at high school,middle level colleges level and at university level.I have been an active academic essays writer since 2011 where I have worked with some of the most reputable essay companies based in Europe and in the US.I have over the years perfected my academic writing skills as a result of tackling numerous different assignments.I do not plagiarize and I maintain competitive quality in all the assignments that I handle.I am driven by strong work ethics and a firm conviction that I should "Do Unto others as I would Like them to do to me".
4.80+
76+ Reviews
152+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Calculate the equilibrium constant for the acid-base reactions between the following pairs of reactants. a. HCl + H2O b. CH3COOH + H2O c. CH3NH2 + H2O CH3NH3 + H20
-
The molecule methylamine (CH3NH2) can act as a monodentate ligand. The following are equilibrium reactions and the thermochemical data at 298 K for reactions of methylamine and en with Cd2+ (aq); (a)...
-
Calculate the standard free-energy change and the equilibrium constant Kp for the following reaction at 25oC. See Appendix C for data. CO(g) + 2H2(g) =CH,OH(g)
-
On 1 January 2022, ABC Company issued 10,000 shares of common stock for $100,000. On 31 December 2022, the company declared and paid dividends of $10,000. Calculate the earnings per share and the...
-
The following data were taken from the records of Nomad Company for March 2008: Administrative expenses ............... $ 58,000 Cost of goods sold (at standard) ............ 885,500 Direct materials...
-
Selected year-end account balances from the adjusted trial balance as of December 31, 2022, for Tippy Corporation is provided below. Instructions a. Prepare closing entries b. Determine the...
-
What conditions must be met for stud mounting an accelerometer?
-
Multiple Choice Question 1. What is a subsidiarys functional currency? a. The parents reporting currency. b. The currency in which transactions are denominated. c. The currency in which the entity...
-
There are many different types of business-to-business consumers/buyers. Discuss different types of customers and describe the differences between them. Your discussion must include an example of...
-
One half-cell in a voltaic cell is constructed from an iron electrode in an Fe(NO 3 ) 2 solution of unknown concentration. The other half-cell is a standard hydrogen electrode. A potential of 0.49 V...
-
One half-cell in a voltaic cell is constructed from a silver wire electrode in a AgNO 3 solution of unknown concentration. The other half-cell consists of a zinc electrode in a 1.0 M solution of...
-
What does the term duration mean to bond investors and how does the duration of a bond differ from its maturity? What is modified duration, and how is it used? What is effective duration, and how...
-
The start of a business requires answers to several complicated questions. One such question is the type of business structure for the new organization. This week we focus on the partnership...
-
Provide academic research and find out what variables affect the insurance premiums and how, against 1) stock market losses (Put option value) 2) mortgage insurance, 3) car insurance, and 4) life...
-
Throughout McMillan's speech, he discusses the idea that there is an essential communication bridge between law enforcement and civilians. The concept of "the bridge" McMillan is talking about is...
-
In your Point, Good Leadership is About Communicating "Why," the author claims that communication presentations require careful planning. She states, "If we present without addressing our audience's...
-
According to Leah Selim, What is gastro-diplomacy? How is it a useful tool for understanding other cultures? What is meant by "Food Rules?" How do they effect college students?
-
Refer to Exercise 16.9. The company has just hired a 22-year-old telemarketer. Predict with 95% confidence how long he will stay with the company.
-
Read the case study Richter: Information Technology at Hungarys Largest Pharma and answer the following question: How does the organization ensure the accuracy of the data it stores?
-
A roofing tile falls from rest off the roof of a building. An observer from across the street notices that it takes 0.43 s for the tile to pass between two windowsills that are 2.5 m apart. How far...
-
You are standing at the top of a deep, vertical cave and want to determine the depth of the cave. Unfortunately, all you have is a rock and a stopwatch. You drop the rock into the cave and measure...
-
Your friend is an environmentalist who is living in a tree for the summer. You are helping provide her with food, and you do so by throwing small packages up to her tree house. If her tree house is...
-
Current debates about legalization of marijuana highlight some of the tensions between state and federal power and authority. After reading the three cases below, how do you make sense of the line...
-
What is the central idea of this article. Howe, Scott W. (2015) "The Implications of Incorporating the Eighth Amendment Prohibition on Excessive Bail," Hofstra Law Review: Vol. 43: Iss. 4, Article 4....
-
What if you are in the car with a friend when you are pulled over by police and the officer draws his gun and tells you and your friend to get out of the car? You know you have done nothing wrong....

Study smarter with the SolutionInn App


