Calculate the binding energy per mole of nucleons for iron-56. Masses needed for this calculation (in g/mol)
Question:
Calculate the binding energy per mole of nucleons for iron-56. Masses needed for this calculation (in g/mol) are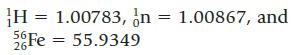
Transcribed Image Text:
H = 1.00783, n = 1.00867, and 56Fe = 55.9349
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
The binding energy per mole of nucleons can be calculated using the mass defect and the Avogadros number The mass defect is the difference between the ...View the full answer

Answered By

Stacy kosgei
I offer quality, original and timely services; Highly credible and void of plagiarism. Your success is my pleasure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Calculate the binding energy per mole of nucleons for calcium-40, and compare your result with the value in Figure 25.4. Masses needed for this calculation are (in g/mol) Data given in Figure 25.4 H...
-
Calculate the binding energy per mole of nucleons for Masses needed for this calculation are 16 8 O.
-
Calculate the binding energy per mole of nucleons for nitrogen-14. The mass of nitrogen-14 is 14.003074.
-
Jilmar Company acquired a delivery truck and made payment of P2,870,000 analyzed as follows: Price of truck - P2,500,000 Charge for extra equipment - P150,000 Value added tax (recoverable) -...
-
Thorup Company sells 5,000 units at $40 per unit. Variable costs are $34 per unit, and fixed costs are $10,000. Determine (a) The contribution margin ratio, (b) The unit contribution margin , and (c)...
-
Planar waves from a monochromatic light source are normally incident on a circular obstacle, which casts a shadow on a screen positioned behind the obstacle. What do the wave properties of light...
-
Explain in laymans terms what the law of the excluded middle means. What difficulties might you encounter in logical deduction if you ignored the law of the excluded middle?
-
Stephen Hall is developing a program in supply chain management certification for managers. Hall has listed a number of activities that must be completed before a training program of this nature...
-
The owner of the All American Hero Restaurant asked his Certified Personal Accountant ( CPA ) to construct a balance sheet for the restaurant. The CPA requested all relevant financial records from...
-
Copper(II) acetate containing 64 Cu is used to study brain tumors. This isotope has a half-life of 12.7 hours. If you begin with 25.0 g of 64 Cu, what mass remains after 63.5 hours?
-
Calculate the binding energy in kilojoules per mole of nucleons of P for the formation of 30 P and 31 P. The required masses (in g/mol) are H = 1.00783, n = 1.00867, P 1.00867, P = 29.97832, 15 and P...
-
Flying Gator Corporation and its 100%-owned subsidiary, T Corporation, have filed consolidated tax returns for many years. Both corporations use the hybrid method of accounting and the calendar year...
-
Brad Rowe was a self-employed window washer earning approximately $700 per week. One day, while cleaning windows on the eighth floor of the First National Bank Building, he tripped and fell from the...
-
Michael Cheung was seriously injured in a snowboarding accident that broke both his legs and an arm. His medical expenses included five days of hospitalization at $900 a day, $6,200 in surgical fees,...
-
Consider an ideal air-standard Brayton cycle operating with inlet temperature (T 1 ) and pressure (P 1 ) of 300 K and 100 kPa, respectively. The heat added per unit mass, q 2 3 , is 1050 kJ/kg....
-
If Barbara Guarin is single and in the 28% tax bracket, calculate the tax associated with each of the following transactions. (Use the IRS regulations for capital gains in effect in 2008.) a. She...
-
Last year Charles and Kathy Price bought a home with a dwelling replacement value of $250,000 and insured it (via an HO-3 policy) for $210,000. The policy reimburses for actual cash value and has a...
-
Cathy Muench a factory worker earns $1,000 each week. In addition, she will receive a $4,000 bonus at year-end and a four-week paid vacation. Prepare the entry to record the weekly payroll and the...
-
Anna, a high school counselor, devised a program that integrates classroom learning with vocational training to help adolescents at risk for school dropouts stay in school and transition to work...
-
Medium fuel oil at 25C is to be pumped at a flow rate of 200 m 3 /h through a DN 125 Schedule 40 pipe over a total horizontal distance of 15 km. The maximum working pressure of the piping is to be...
-
Glycerin at 25C flows through a straight hydraulic copper tube, 80 mm OD 2.8 mm wall, at a flow rate of 180 L/min. Compute the pressure difference between two points 25.8 m apart if the first point...
-
A 3-in Schedule 40 steel pipe is 5000 ft long and carries a lubricating oil between two points A and B such that the Reynolds number is 800. Point B is 20 ft higher than A. The oil has a specific...
-
Focusing on the sector of education and the popular tool of AI Chat GPT, identify the positive and negative effects that tool could have for the educational system based on expert analysis and give...
-
Consider following payoff matrix for two firms Firm 2 High Price Firm 1 High Price 34,60 Low Price 75,25 If Firm 1 follows a maximin strategy, it would choose Low Price 21,90 22,18
-
Assess whether a business is monopolistic is by looking at its domination; both market-wise and structure-wise. Evaluate the market domination in Malaysia by providing THREE (3) examples from...

Study smarter with the SolutionInn App


