Calculate the molar mass of each hydrated compound. The water of hydration is included in the molar
Question:
Calculate the molar mass of each hydrated compound. The water of hydration is included in the molar mass.
Given Data
 (a). Ni(NO3)2 ∙ 6 H2O
(a). Ni(NO3)2 ∙ 6 H2O
(b). CuSO4 ∙ 5 H2O
Transcribed Image Text:
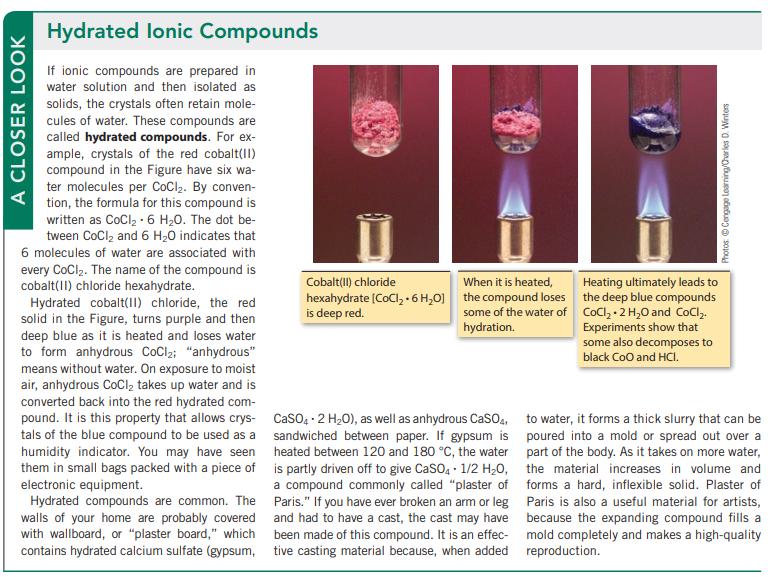
A CLOSER LOOK Hydrated lonic Compounds If ionic compounds are prepared in water solution and then isolated as solids, the crystals often retain mole- cules of water. These compounds are called hydrated compounds. For ex- ample, crystals of the red cobalt(II) compound in the Figure have six wa- ter molecules per CoCl₂. By conven- tion, the formula for this compound is written as CoCl₂-6 H₂0. The dot be- tween CoCl₂ and 6 H₂O indicates that 6 molecules of water are associated with every CoCl₂. The name of the compound is cobalt(II) chloride hexahydrate. Hydrated cobalt(II) chloride, the red solid in the Figure, turns purple and then deep blue as it is heated and loses water to form anhydrous CoCl₂; "anhydrous" means without water. On exposure to moist air, anhydrous CoCl₂ takes up water and is converted back into the red hydrated com- pound. It is this property that allows crys- tals of the blue compound to be used as a humidity indicator. You may have seen them in small bags packed with a piece of electronic equipment. Hydrated compounds are common. The walls of your home are probably covered with wallboard, or "plaster board," which contains hydrated calcium sulfate (gypsum, Cobalt(II) chloride When it is heated, hexahydrate (CoCl₂ + 6H₂O] the compound loses is deep red. some of the water of hydration. CaSO4 2 H₂O), as well as anhydrous CaSO4, sandwiched between paper. If gypsum is heated between 120 and 180 °C, the water is partly driven off to give CaSO4 1/2 H₂O, a compound commonly called "plaster of Paris." If you have ever broken an arm or leg and had to have a cast, the cast may have been made of this compound. It is an effec- tive casting material because, when added Photos Cengage Learning/Charles D. Winters Heating ultimately leads to the deep blue compounds CoCl₂.2 H₂O and CoCl₂. Experiments show that some also decomposes to black CoO and HCI. to water, it forms a thick slurry that can be poured into a mold or spread out over a part of the body. As it takes on more water, the material increases in volume and forms a hard, inflexible solid. Plaster of Paris is also a useful material for artists, because the expanding compound fills a mold completely and makes a high-quality reproduction.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To calculate the molar mass of a hydrated compound we need to add the molar mass of each element in ...View the full answer

Answered By

Mary Boke
As an online tutor with over seven years of experience and a PhD in Education, I have had the opportunity to work with a wide range of students from diverse backgrounds. My experience in education has allowed me to develop a deep understanding of how students learn and the various approaches that can be used to facilitate their learning. I believe in creating a positive and inclusive learning environment that encourages students to ask questions and engage with the material. I work closely with my students to understand their individual learning styles, strengths, and challenges to tailor my approach accordingly. I also place a strong emphasis on building strong relationships with my students, which fosters trust and creates a supportive learning environment. Overall, my goal as an online tutor is to help students achieve their academic goals and develop a lifelong love of learning. I believe that education is a transformative experience that has the power to change lives, and I am committed to helping my students realize their full potential.
5.00+
4+ Reviews
22+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Calculate the molar mass of each hydrated compound. Note that the water of hydration is included in the molar mass. Given Data (a). H 2 C 2 O 4 2 H 2 O (b). MgSO 4 7 H 2 O, Epsom salt A CLOSER LOOK...
-
Elaborate figures given below Price per pair (dollars) 105 90 75 60 45 30 15 0 10 20 30 I E Equilibrium IS 40 50 60 70 Quantity of tennis shoes (thousands of pairs per year) 80 90 100 D
-
d^w Consider a beam that can be described by Euler-Bernoulli Beam ODE EI q(x). Assume the beam 10 meters long, has an area moment of inertial of 8e-5 m, has one fixed-end boundary condition (w' = 0...
-
When a company's bookkeeper started to prepare the monthly bank reconciliation, the cash account showed a balance of P528.600. At the end of the month, the following information was available from...
-
Your supervisor, Jesse Baker, has asked you to begin working on data design tasks for the new information system, which will be implemented as a relational database. You will need to identify the...
-
After the dot-com business he tried to start folded, David "Marty" Dawkins decided to pursue his boyhood dream of owning a bike factory. After several false starts, he finally got the small company...
-
With reference to the previous exercise, construct a 90% confidence interval for the true population mean quantity of gravel in concrete mixes. Data From Previous Exercise 7.1 A construction engineer...
-
The Red Division of Tarkington Company reported the following data for the current year. Sales .......... $3,000,000 Variable costs ...... 1,950,000 Controllable fixed costs .. 600,000 Average...
-
As a quality manager , you will be continuously reviewing your facility against best practices and benchmarks in the industry. What are some key indicators used in health care to determine quality?...
-
Analysis of a 10.0-g sample of apatite (a major component of tooth enamel) showed that it was made up of 3.99 g Ca, 1.85 g P, 4.14 g O, and 0.020 g H. List these elements based on relative amounts...
-
You are given 0.10-g samples of K, Mo, Cr, and Al. List the samples in order of the amount (moles), from smallest to largest.
-
Explain the relevance of managers using shaping behavior.
-
Consider the same simple linear regression (SLR) model Gauss Markov assumptions: Y = Bo + B X + u, where the error term u has an unconditional variance of o. Your friend highly recommends this...
-
25 lbm of water with a quality value of 0.55 exists in a rigid tank at 80F. Determine the a. The pressure [psia] of the water in the tank (5 points) b. The volume [ft] of the tank (15 points)
-
Discuss the influence of Stuart Hall in the development of the field of cultural studies.
-
For the beam below (see dimensions below); (1) Calculate, dimension and sketch the location of the horizontal centroidal axis (2) Calculate the Second Moment of Area Ixx (horizontal axis only) (3)...
-
The investor buys 100 shares of stock for $10 a share and sells it for $14. Also assume the margin requirement is 60%, the commission rate is 5% of the purchase or sale price, the interest rate is...
-
A firm that has no debt has a market value of $100 million and a cost of equity of 11%. In the MillerModigliani world. a. what happens to the value of the firm as the leverage is changed (assume no...
-
Use the method of Example 4.29 to compute the indicated power of the matrix. 1 0 1
-
Draw the product(s) obtained from hydroboration-oxidation of (E)-3-methyl-3-hexene.
-
The products obtained from hydroboration-oxidation of cis-2-butene are identical to the products obtained from hydroboration-oxidation of trans-2-butene. Draw the products and explain why the...
-
As the pressure is increased at 45C, ice I is converted to ice II. Which of these phases has the lower density?
-
Solve the equation. 2 x 1 5 + 4 x 5 = 1 4
-
Solve the equation. x - 3 5 + 3 5 = x 6
-
Find all the values of x such that sin ( 2 x ) = sin ( x ) and 0 less than equal to x less than equal to 2 pi ( enter your answers as a comma - separated list. )

Study smarter with the SolutionInn App


