Complete and balance the equations for the following acidbase reactions. Name the reactants and products. (a) H3PO4(aq)
Question:
Complete and balance the equations for the following acid–base reactions. Name the reactants and products.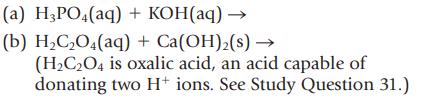
Transcribed Image Text:
(a) H3PO4(aq) + KOH(aq) → (b) H₂C₂O4(aq) + Ca(OH)₂(s) → (H₂C₂O4 is oxalic acid, an acid capable of donating two H+ ions. See Study Question 31.)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a H3PO4aq KOHaq In this reaction phosphoric acid H3PO4 reacts with potassium hydroxid...View the full answer

Answered By

Muhammad Salman Alvi
Well, I am a student of Electrical Engineeing from Information Technology University of Punjab. Just getting into my final year. I have always been good at doing Mathematics, Physics, hardware and technical subjects. Teaching profession requires a alot of responsibilities and challenges.
My teaching experience started as an home tutor a year ago. When I started teaching mathematics and physic subjects to an O Level student. He was about 14 years old. His name was Ibrahim and I used to teach him for about 2 hours daily. Teaching him required a lot of patience but I had to be polite with him. I used to give him a 5 min break after 1 hour session. He was quite weak in basic maths and calculation. He used to do quite a lot of mistakes in his homework which I gave him weekly. So I decided to teach him basics from scratch. He used to say that he got the concept even if he didn't. So I had to ask him again and again. I worked on his basics for a month and after that I started taking a weekly test sesions. After few months he started to improve gradually. Now after teaching him for about a year I can proudly say that he has improved alot. The most important thing was he managed to communicate all the difficullties he was facing. He was quite capable and patient. I had a sincere desire to help him reach to its full potential. So I managed to do that. We had a very good honest relationship of a student and a teacher. I loved teaching him as a tutor. Now having an experience of one year teaching I can read students quite well. I look forward to work as an online tutor who could help students in solving their all sort of difficulties, problems and queries.
4.90+
29+ Reviews
43+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Complete and balance the equations for the following reactions. a. Li(s) + N2(g) b. Rb(s) + S(s) c. Cs(s) + H2O(l) d. Na(s) + Cl2(g)
-
Complete and balance the equations for the following acidbase reactions. Name the reactants and products. (a) CH3COH(aq) + Mg(OH)(s) (b) HClO4(aq) + NH3(aq)
-
Complete and balance the following nuclear equations by supplying the missing particle: 98 53 59 0
-
A 12-year bond has an annual coupon rate of 9%. The par value of the bond is $1,000 and the bond has a yield to maturity of 7%. Which of the following statements is correct? If the market interest...
-
Can you think of reasons why a government might be concerned about a large current account deficit or surplus? Why might a government be concerned about its official settlements balance (that is, its...
-
In Hawaii shark attacks are very rare. Furthermore, about 55% of all shark attacks are serious, but not fatal (Reference: Sharks of Hawaii, Dr. Leighton Taylor, University of Hawaii Press). You are...
-
Fuel injector is used in (a) S.I. engine (b) C.I. engine (c) Gas engine (d) Petrol engine
-
Air at 38C and 97% relative humidity is to be cooled to 18C and fed into a plant area at a race of 510m3/min. (a) Calculate the rate (kg/mm) at which water condenses. (b) Calculate the cooling...
-
Disney needs to close Magic Kingdom to remodel and update the castle. Disney's advisors presented two renovation alternatives: (1) a quick facelift or (2) a complete rebuild. The quick facelift would...
-
Write a balanced equation for the reaction of sulfur trioxide gas with water.
-
Write a balanced equation for reaction of the basic oxide, magnesium oxide, with water.
-
Using the central bank balance sheet diagrams, evaluate how each of the following shocks affects a countrys ability to defend a fixed exchange rate. In the following answers, we assume the central...
-
Before recording adjusting entries on December 31, the Store Supplies account had a $1,900 debit balance, while a physical count of the supplies showed $300 of unused supplies on hand. Prepare the...
-
How many times will each account on the trial balance be used within the financial statements?
-
The pre-qualifying and qualifying stages of the personal selling process are very important. These stages help sales professionals identify and focus their time, money and efforts on potential...
-
How is the preparation of the statement of cash flow different from other financial statements as it relates to the source of information?
-
K One hundred college seniors attending a career fair at a university were categorized according to gender and according to primary career motivation. The table below shows the results. If one of...
-
Detner International purchases 80% of the outstanding stock of Hardy Company for $1,600,000 on January 1, 2015. At the purchase date, the inventory, equipment, and patents of Hardy Company have fair...
-
Chicago Company sold merchandise to a customer for $1,500 cash in a state with a 6% sales tax rate. The total amount of cash collected from the customer was $558. $600. $642. $636. Nevada Company...
-
Identify the reagents necessary to accomplish each of the transformations below: (a) (b)
-
Predict the major product of each reaction below: (a) (b) (c) RCO3H RCO, ? H RCO3H
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) (d) (e) (f) (g) -OH
-
A company has just announced a dividend of $0.80 for this year and $0.835 for the next year. Dividends are expected to grow at a constant rate indefinitely. What is the current stock price if the...
-
Respond using research from Journal Articles to validate your response. Discuss the laws that govern wages, pensions, and employee compensation in the USA.
-
What are some of the biggest challenges and changes in trends for Human Resources due to the COVID-19 pandemic which has organizations in flux since 2020 especially with employees working remotely?

Study smarter with the SolutionInn App


