Complete the following nuclear equations. Write the mass number and atomic number for the remaining particle, as
Question:
Complete the following nuclear equations. Write the mass number and atomic number for the remaining particle, as well as its symbol.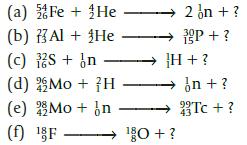
Transcribed Image Text:
(a) Fe + He 2 on + ? (b) Al + He 3P + ? (c) 2S + n →→→→H + ? (d) Mo + H →in + ? (e) Mo + n →→→→Tc+? 43 (f) F ¹0+?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)

Answered By

Shristi Singh
A freshman year metallurgy and material science student in India.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Complete the following nuclear equations and identify X in each case: 135
-
Consider the following depictions of two atoms, which have been greatly enlarged so you can see the subatomic particles. a. How many protons are present in atom A? b. What is the significance of the...
-
Complete the following nuclear decay equations by filling in the blanks: + 139
-
If M is the midpoint of XY, find the coordinates of Y when X and M have the following coordinates: X(-4,2), M(0,3) Please write formulas too
-
Snowflake Flour Company manufactures flour by a series of three processes, beginning in the Milling Department. From the Milling Department, the materials pass through the Sifting and Packaging...
-
The common stock of Buildwell Conservation & Construction, Inc. (BCCI), has a beta of .8. The Treasury bill rate is 4% and the market risk premium is estimated at 7%. BCCI's capital structure is 30%...
-
Use the data set, which represents the ages of 30 executives. Which ages are below the 75th percentile? 43 57 65 47 57 41 56 53 61 54 56 50 66 56 50 61 47 40 50 43 54 41 48 45 28 35 38 43 42 44
-
Johnson, Larson, and Kragen own an advertising agency that they operate as a partnership. The partnership agreement includes the following: a. Johnson receives a salary of $50,000. b. Larson receives...
-
How would you respond to the following? (include questions for each response) Statement 1: Hi Destinee. Good work here, and thank you for your contributions to this thread. In hospitality,...
-
Complete the following nuclear equations. Write the mass number, atomic number, and symbol for the remaining particle. (a) Be + ? (b) ? + in (c) Ca + ? (d) 2 Am + He 96 (e) 246Cm + C (f) 23 U + ? Li...
-
Oxygen-15 is used in the medical procedure called positron emission tomography. This isotope is prepared on a cyclotron from irradiation of nitrogen-14 with deuterium nuclei. It decays to nitrogen-15...
-
Following the directions discussed in this chapter, write to the direct marketing association and the three credit bureaus and request to opt out of preapproved credit card mailings. Turn in a copy...
-
Read the poem by Sherman Alexie entitled, The Powwow at the End of the World (1996). The Powwow at the End of the World and answers the questions. 1. who is speaking 2. what is the speaker speaking...
-
A topic that has a local focus, such as a change that needs to happen in your community. Please include three things: what the need for change is, what your planned solution is, and who the audience...
-
Suggest which solution to a gender-neutral pronoun you think would be the best option and why. What do you think is the best approach to appeal to the Real Academia Espaola to make them official...
-
Please take a look at the Checklist below and talk about your thoughts on what it recommends compared to your usual habits when it comes to drafting and revising your writing. Feel free to address...
-
Important considerations pertaining to the amount of inventory a business has available to sell, allows for discovering what items are hot sells for a business. Knowing this you will know how much...
-
Exercise described a survey of adults wherein, on the basis of several probing questions, each was classified as either a member of the health conscious group (code = 1) or not (code = 2) and whether...
-
The sales department of P. Gillen Manufacturing Company has forecast sales in March to be 20,000 units. Additional information follows: Finished goods inventory, March 1 . . . . . . . . . . . . . . ....
-
Consider the motion of a bicycle with air drag included. We saw how to deal with the motion on a hill in connection with Figure 4.33. Now assume the bicycle is coasting on level ground and is being...
-
A skier travels down a steep, friction less slope of angle 20 with the horizontal. Assuming she has reached her terminal velocity, estimate her speed.
-
A soccer ball has a diameter of about 22 cm and a mass of about 430 g. What is its terminal velocity?
-
1. Nail & Hammer Shack purchased 246 table fans of a particular make. The unit cost was $29.50 and the regular selling price was $60.00 each. The store sold 122 of them at the regular selling price....
-
In its annual report to stockholders, Hakobe Inc. presents a condensed balance sheet with detailed data provided in supplementary schedules. 1. From the adjusted trial balance of Hakobe, prepare the...
-
You are given the stockholders' equity section of Smart Corporation on 31-12-2024, with some missing information, as follows: Stockholders' Equity: Paid in Capital: Capital stock: 8%, $100 par,...

Study smarter with the SolutionInn App


