Cuprite is a semiconductor. Oxide ions are at the cube corners and in the cube center. Copper
Question:
Cuprite is a semiconductor. Oxide ions are at the cube corners and in the cube center. Copper ions are wholly within the unit cell.
(a) What is the formula of cuprite?
(b) What is the oxidation number of copper?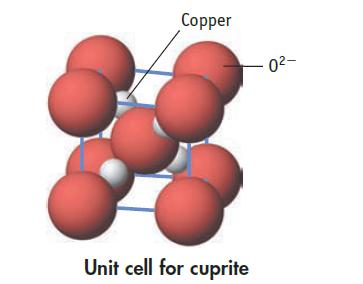
Transcribed Image Text:
Copper Unit cell for cuprite 0²-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a The formula of cuprite is Cu2O This is because there are two copp...View the full answer

Answered By

Usman Nasir
I did Master of Commerce in year 2009 and completed ACCA (Association of Chartered Certified Accountants) in year 2013. I have 10 years of practical experience inclusive of teaching and industry. Currently i am working in a multinational company as finance manager and serving as part time teacher in a university. I have been doing tutoring via many sites. I am very strong at solving numerical / theoretical scenario-based questions.
4.60+
16+ Reviews
28+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
A particular unit cell is cubic with ions of type A located at the corners and facecenters of the unit cell and ions of type B located at the midpoint of each edge of the cube and at the...
-
Classify each of the following solids as an metallic, molecular, network or ionic solids : Silver (Ag), Silicon Carbide (SiC), Silicon (Si), white phosphorous (P 4 ) Glucose (C 6 H 12 O 6 ),...
-
The structures of another class of high-temperature ceramic superconductors are shown below. a. Determine the formula of each of these four superconductors. b. One of the structural features that...
-
8) Implement a class UserNames which requires an ArrayList of names (user names), consider the following static methods methods: initilizeUserName(), deleteUserName(), addUserName() and display...
-
Evaluate this statement: Good customer service doesnt do you much good, but poor customer service can kill you. Missionary or supporting salespeople do not normally try to secure orders. They are...
-
Estimate the area in each figure shown in Problems 39-46. y A 5 5432 4 1 -0.51 L -2 1.0 2.0 3.0x
-
Tennessee law imposes durational-residency requirements on persons and companies wishing to operate retail liquor stores, requiring applicants for an initial license to have resided in the state for...
-
The comparative statements of Corbin Company are presented below and on page 884. Additional data: The common stock recently sold at $19.50 per share. Instructions Compute the following ratios for...
-
Outback Outfitters sells high quality hiking boots. They have a capacity to make1500 pairs per year. Each pair of hiking boots sells for $630. Annual fixed costs are $280,000,and the variable costs...
-
The mineral fluorite, which is composed of calcium ions and fluoride ions, has the unit cell shown here. (a) What type of unit cell is described by the Ca 2+ ions? (b) Where are the F ions located,...
-
Rutile, TiO 2 , crystallizes in a structure characteristic of many other ionic compounds. How many formula units of TiO 2 are in the unit cell illustrated here? (The oxide ions marked by an x are...
-
Allen has taxable income of \(\$ 75,475\) for 2018. Using the Tax Rate Schedules in the Appendix, compute Allen's income tax liability before tax credits and prepayments for each of the following...
-
A 0.30T magnetic field points in the axis. A 1.5 C charge enters the region with a speed of 4.0m/s making a 30, degree angle with the x-axis. What is the magnitude of the force experienced by the...
-
1) A trader entered into a long S&P index futures contract when the futures price is 3,112 and closed it out when the futures price is 3,075. What is his gain or loss in dollars? The contract...
-
ABC Consider an economy with two industries, using a common resource to produce two different types of goods and to achieve a certain level of technology. Given a fixed technology level K (considered...
-
What are temporary earnings? Explain.
-
Propose and discuss the potential capital structure-related impact of government response to the corporate scandals during 2000 and 2003 and the financial crisis during 2008 and 2009. Discuss the...
-
Tusk Company has 60,000 shares of its $1 par value common stock outstanding. Prepare journal entries for the following transactions as they relate to the companys common stock : July 17 Declared a 10...
-
A manufacturer can sell product 1 at a profit of $20 per unit and product 2 at a profit of $40 per unit. Three units of raw material are needed to manufacture one unit of product 1, and six units of...
-
Draw the alkyl halide that would be necessary to make the amino acid tyrosine using an amidomalonate synthesis. This alkyl halide is highly susceptible to polymerization. Draw the structure of the...
-
When leucine is prepared with an amidomalonate synthesis, isobutylene (also called 2methylpropene) is a gaseous byproduct. Draw a mechanism for the formation of this byproduct.
-
The side chain of tryptophan is not considered to be basic, despite the fact that it possesses a nitrogen atom with a lone pair. Explain.
-
A 6-month bank loan of $1,000.00 required the equal payments monthly. After three payments were made, the borrower decided to pay off the loan at the 4th month before he paid his monthly payment. The...
-
Market Dynamics (10 Points) Between 1980 and 1990, the number of employed women grew sharply but the number of women employed as secretaries actually fell. There are two theories as to why this...
-
3. If the policy proposed in question 2 is adopted, analyze the potential effects of this policy on the demand for workers under the age of 65. That is, if employers who want their employees to be...

Study smarter with the SolutionInn App


