Explain why benzenesulfonic acid is a Brnsted acid. H-0: :0-S :0: benzenesulfonic acid
Question:
Explain why benzenesulfonic acid is a Brønsted acid.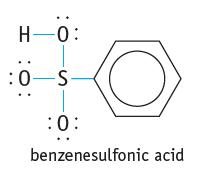
Transcribed Image Text:
H-0: :0-S :0: benzenesulfonic acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Benzenesulfonic acid is a Brnsted acid because it can donate a proton H to another substa...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Work Problem 3.27 for the reverse reactions in Fig. P3.27 Problem 3.27 (a) Classify each reaction step in the forward direction with one or more of the following terms: (1) A Lewis acid-base reaction...
-
Benzenesulfonic acid is a monoprotic acid with pKa = 2.25. Calculate the pH of a buffer composed of 0.150 Mbenzenesulfonic acid and 0.125 M sodium benzenesulfonate.
-
Write a stepwise mechanism for the hydrolysis of chlorobenzene under the conditions shown in Table 24.3. TABLE 24.3 Industrial Syntheses of Phenol Reaction and comments Chemical equation Reaction of...
-
Since the early 2000s, there has been a significant increase in the price of corn-based ethanol. a. A key input in the production of corn-based ethanol is corn. Use an appropriate diagram to explain...
-
In the past decade, the Japanese economy has fallen from its lofty levels of the 1980s. As a consequence, many Japanese companies have been forced to downsize. In most companies, one of two...
-
Campus Theater adjusts its accounts every month. The companys unadjusted trial balance dated August 31, 2015, is on page 176. Additional information is provided for use in preparing the companys...
-
If the assembly fits snugly between the two supports A and C when the temperature is T 1 , determine the normal stress developed in both segments when the temperature rises to T 2 . Both segments are...
-
Lone Mountain Extraction, which mines ore in Idaho, uses a calendar year for both financial-reporting and tax purposes. The following selected costs were incurred in December, the low point of...
-
Larry Culp Redefines Success at GE When Larry Culp became the 12th chief executive officer of General Electric, he was the first CEO to be brought in from outside the company, and GE desperately...
-
m-Nitrophenol, a weak acid, can be used as a pH indicator because it is yellow at a pH above 8.6 and colorless at a pH below 6.8. If the pH of a 0.010 M solution of the compound is 3.44, calculate...
-
The chapter opening photograph (page 670) showed how the cobalt(II) chloride equilibrium responded to temperature changes. (a) Look back at that photograph. Is the conversion of the red cation to the...
-
The accounts of Sunny Stream Service, Inc., at March 31, 2010, are listed in alphabetical order. Requirements 1. All adjustments have been journalized and posted, but the closing entries have not yet...
-
The insurance company accountant must apply what set of accounting principles when creating insurer financial statements for the state regulators?
-
The human resources department of Matrix International Corporation (MIC) has recently hired you to revamp its performance appraisal system. The present performance appraisal system has not been...
-
What are the legislative, regulatory and industry requirements that must be met with in relation to financial reporting for this company for the year ended 30 June 2022?
-
Where does Management use Non-GAAP measures? Explain.
-
How do non-linear dynamics influence human performance and decision-making in organizational settings? Explain
-
In theory, Republicans are more supportive of free enterprise. Do the data allow us to infer that Republican Party supporters are more likely to work for themselves (WRKSLF: 1 = Work for themselves,...
-
CLASS PERIO Solving Linear Equations: Variable on Both Sides Solve each equation. 1) 6r+ 7 = 13 + 7r 3) -7x-3x+2=-8x-8 5)-14 +66+7-26=1+5b 7) n-3n = 14-4n 2) 13-4x=1-x 4)-8-x= x - 4x 6)n+2=-14-n 8)...
-
A certain metal surface emits electrons when light is shone on it. (a) How can the number of electrons per second be increased? (b) How can the energies of the electrons be increased?
-
Would you expect the gravitational attractive force between two protons in a nucleus to counterbalance their electrical repulsion? Calculate the ratio between the electric and gravitational forces...
-
Which constituents of an atom consist of quarks and which do not?
-
A thrill-seeker (mass 80.0 kg) bungee jumps off a bridge spanning a 110 m deep river canyon. After they have fallen 70.0 m the cord begins stretching, bringing them to rest 10.0 m above the river. a....
-
You are a stunt person for a major blockbuster movie. In one scene you are driving a bright yellow Camaro trying to escape a rampaging robot. You are supposed to slam on the brakes and just barely...
-
You work for a company that designs bulletproof vests for law enforcement officers. The vest you are working on is being designed to stop a 20.0 0.1 g bullet traveling at 200 3 m/s when it is fired...

Study smarter with the SolutionInn App


