Give approximate values for the indicated bond angles. (a) OSO angle in SO 2 (b) FBF angle
Question:
Give approximate values for the indicated bond angles.
(a) O—S—O angle in SO2
(b) F—B—F angle in BF3
(c) Cl—C—Cl angle in Cl2CO
(d) H—C—H (angle 1) and C—C≡N (angle 2) in acetonitrile
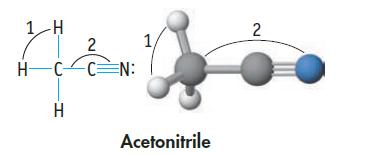
Transcribed Image Text:
1 H HC H 2 CN: 1, Acetonitrile 2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a OSO angle in SO2 The OSO angle in SO2 is approximately 120 degrees This is because the sulfur atom in SO2 is sp2 hybridized which means that it has three sp2 hybrid orbitals and one unhybridized p o...View the full answer

Answered By

Aqib Parvej
I am teaching since my graduation time so I have teaching experience of about 5 years and in these years I learn to teach in the best and interesting way .
4.80+
20+ Reviews
41+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Give the approximate values for the indicated bond angles in the following molecules: (a) (b) (c) (d) 4 3 6 H-COCH 7 H
-
How has the war in Ukraine impacted the U.S. financial system and economy (consider the impact on international portfolio investment vehicles and the following questions in your response)? Why have...
-
Give approximate values for the indicated bond angles: (a) ClSCl in SCl 2 (b) NNO in N 2 O (c) Bond angles 1, 2, and 3 in vinyl alcohol (a component of polymers and a molecule found in outer space) 1...
-
is an acute angle such that tan() =1/3. What is the value of 1010. (sin + cos)? 3.
-
Describe the benefits of real-time data warehousing at Continental. Continental Airlines was founded in 1934 with a single engine Lockheed aircraft on dusty runways in the American Southwest. Over...
-
A granular material is observed being dumped from a conveyor belt. It forms a conical pile with about the same slope angle, 1.8 horizontal to 1 vertical. What is the angle of internal friction of...
-
Water flows in a constant-diameter pipe with the following conditions measured: At section (a) \(p_{a}=32.4 \mathrm{psi}\) and \(z_{a}=56.8 \mathrm{ft}\); at section (b) \(p_{b}=29.7 \mathrm{psi}\)...
-
What will your portfolio be worth in 10 years? In 20 years? When can you stop working? The Human Resources Department at Four Corners Corporation was asked to develop a financial planning model that...
-
x- y 5. (5) Let f(x, y) = a. Sketch the level curve of f where z = 0. b. Use one path to come up with a conjecture for the following limit and then use the epsilon-delta definition of a limit to...
-
The following molecules or ions all have three oxygen atoms attached to a central atom. Draw a Lewis structure for each one, and then describe the electron-pair geometry and molecular geometry around...
-
The following molecules or ions all have two oxygen atoms attached to a central atom. Draw a Lewis structure for each one, and then describe the electron-pair geometry and molecular geometry around...
-
For any distribution with mean m 0 and standard deviation , the ratio /m is called a coefficient of variation (c.v.). (a) How would you interpret the situation when the c.v. of one r.v. is larger...
-
Why have smaller commercial banks been able to survive alongside larger banks?
-
How did the financial crisis of the 1930s contribute to the Great Depression? How did policymakers deal with this situation?
-
Contrast the features of a mutual fund investment with those of retail deposits at commercial banks or other depository institutions. In particular, what are their differences in risk and return? Why...
-
What is meant by maturity transformation? How is this related to interest rate risk? How can depository institutions reduce interest rate risk? How does maturity transformation lead to vulnerability...
-
When net worth of financial institutions drops because asset prices fall, how does leverage of these institutions compound the need to cut back on lending? How is a credit crunch involved?
-
How are property, plant, and equipment accounts valued on the balance sheet? Why?
-
If a and b are positive numbers, find the maximum value of f ( x ) = x a (9 x ) b on the interval 0 x 9.
-
A 1 H 19 F molecule, with a bond length of 91.68 pm, absorbed on a surface rotates in two dimensions. a. Calculate the zero point energy associated with this rotation. b. What is the smallest quantum...
-
Verify that 1 (x) in Equation 18.31 is a solution of the Schrdinger equation for the quantum harmonic oscillator. Determine the energy eigenvalue.
-
Evaluate the average kinetic and potential energies, E kinetic and E potential , for the second excited state (n = 2) of the harmonic oscillator by carrying out the appropriate integrations.
-
Problem 2-24 (Algo) (LO 2-6a, 2-6b, 2-8) The following book and fair values were available for Westmont Company as of March 1. Inventory Land Book Value Fair Value $ 587,750 $ 555,25 794,250...
-
Pollachek Co. purchased land as a factory site for $450,000. The process of tearing down two old buildings on the site and constructing the factory required 6 months. The company paid $42,000 to raze...
-
Derrick Wells decided to start a dental practice. The first five transactions for the business follow. 1. Derrick invested $85,000 cash in the business. 2. Paid $25,000 in cash for equipment. 3....

Study smarter with the SolutionInn App


