In the photographic developing process, silver bromide is dissolved by adding sodium thiosulfate. If you want to
Question:
In the photographic developing process, silver bromide is dissolved by adding sodium thiosulfate. 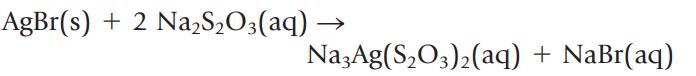
If you want to dissolve 0.225 g of AgBr, what volume of 0.0138 M Na2S2O3, in milliliters, should be used?
Transcribed Image Text:
AgBr(s) + 2 Na,S,O3(aq) Na3Ag(S₂O3)₂(aq) + NaBr(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To calculate the volume of 00138 M Na2S2O3 needed to dissolve 0225 g of AgBr we can use the foll...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
The newspaper production process has come a long way from the old days when the paper was written, edited, typeset and ultimately printed in one building with the journalists working on the upper...
-
Please help with the discusin questions ! I give thumbs up Case #1: Hailing a New Era: Haier in Japan As one of the most valuable brands in China, Haier designs,manufactures, and sells various home...
-
You have $200 and are thinking about betting on the Big Game next Saturday. Your team, the Golden Boars, are scheduled to play their traditional rivals the Robber Barons. It appears that the going...
-
Gabriele Enterprises has bonds on the market making annual payments, with seven years to maturity, a par value of $1,000, and selling for $974. At this price, the bonds yield 7.2 percent. What must...
-
Explain the use of p-charts and c-charts. When would you use one rather than the other? Give examples of measurements for both p-charts and c-charts.
-
When thorium (atomic number 90) decays by emitting an alpha particle, what is the atomic number of the resulting nucleus?
-
In a two-dimensional, incompressible flow field, the \(x\) component of velocity is given by the equation \(u=2 x\). (a) Determine the corresponding equation for the \(y\) component of velocity if...
-
Jay Sports Equipment Company, Inc. incurred the following costs during 20x2. Direct material used.................................................................. $226,200 Direct...
-
What are the challenges and strategies for implementing TPM in highly regulated industries, such as pharmaceuticals or aerospace? How can TPM contribute to compliance and quality assurance in these...
-
You can dissolve an aluminum soft drink can in an aqueous base such as potassium hydroxide. If you place 2.05 g of aluminum in a beaker with 185 mL of 1.35 M KOH, will any aluminum remain? What mass...
-
Hydrazine, N 2 H 4 , a base like ammonia, can react with sulfuric acid. What mass of hydrazine reacts with 250. mL of 0.146 M H 2 SO 4 ? 2 NH(aq) + HSO4(aq) 2 NH5+ (aq) + SO4- (aq)
-
Throughout its history what various strategies did Kodak employ? What was Kodak's strategy prior to 2012?
-
Theories of personality are based on observations of how humans develop behavior patterns. Psychological testing assumes there are certain fixed aspects of human personality and those aspects can be...
-
An investment is being evaluated that will provide equivalent annual cash flows of $1,284 starting 1 year from today, and lasting forever. What is the present value of this cash flow stream if...
-
1. [15] A new product is being developed by a food manufacturer. Depending on the quality of raw materials, different yields are expected, and the quality of the final products will also vary...
-
Why do we need fog lights fitted to a vehicle and why they are mounted and aimed low, and parallel to the road?
-
Most people do not know what you mean by "Geo-Politics". Whilst explaining the meaning of "Geo-Politics", take a real-life Geo-Political issue of your choice occurring in Qatar, and discuss the...
-
What type of account must the sum of all subsidiary accounts be equal to?
-
Consider the setup in Problem 16. Show that the relative speed of the ball and the point of contact on the stick is the same before and immediately after the collision. (This result is analogous to...
-
The average heat evolved by the oxidation of foodstuffs in an average adult per hour per kilogram of body weight is 7.20 kJ kg 1 hr 1 . Assume the weight of an average adult is 62.0 kg. Suppose the...
-
Calculate S, S total , and S surroundings when the volume of 150. g of CO initially at 273 K and 1.00 bar increases by a factor of two in a. An adiabatic reversible expansion b. An expansion against...
-
The maximum theoretical efficiency of an internal combustion engine is achieved in a reversible Carnot cycle. Assume that the engine is operating in the Otto cycle and that C V ,m = 5/2 R for the...
-
Your friend hasn't had a job in over 5 years, and took many pauses in - between jobs before that. He needs help choosing what format to write his resume in for his next job application. What do you...
-
Agrabah Company on December 31 has cash of $18,000, noncash assets of $109,140, liabilities of $92,000, and the following capital balances: Jafar $15,590 and Jasmine $19,550. The firm is liquidated,...
-
What is the market value of one common share? Use the dividend discount model and make the following assumptions when solving this problem: A dividend of $2.50 at time period zero An 18% required...

Study smarter with the SolutionInn App


