In the presence of oxgyen and acid, two half-reactions responsible for the corrosion of iron are Calculate
Question:
In the presence of oxgyen and acid, two half-reactions responsible for the corrosion of iron are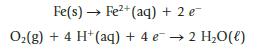
Calculate the the standard potential, E°, and decide whether the reaction is product-favored at equilibrium. Will decreasing the pH make the reaction less thermodynamically product-favored at equilibrium?
Transcribed Image Text:
Fe(s)→ Fe²+ (aq) + 2 e O₂(g) + 4 H+ (aq) + 4 e → 2 H₂O(l)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Calculation of the standard potential E The standard potential of a reaction is calculated by subtra...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
In the presence of the enzyme aconitase, the double bond of aconitic acid undergoes hydration. The reaction is reversible, and the following equilibrium is established: (a) The major tricarboxylic...
-
In a study to compare two different corrosion inhibitors, specimens of stainless steel were immersed for four hours in a solution containing sulfuric acid and a corrosion inhibitor. Forty-seven...
-
In the presence of oxygen and water, two half-reactions responsible for the corrosion of iron are Calculate the the standard potential, E, and decide whether the reaction is product-favored at...
-
If the appropriate discount rate for the following cash flows is 7.13 percent per year, what is the present value of the cash flows? Year Cash Flow 1 ......................$1,400 2...
-
Dayton Industrial produces a variety of chemicals that are used in an array of commercial applications. One popular product, a chemical solvent, contains two very caustic acids, A and B, each of...
-
During 2019, Mount Remarkable Golf Club received $180 000 for membership fees. The accountant credits Unearned Membership Fees, a liability account, for the full amount when cash is received. At 30...
-
The following data are for Huang Wong Ping Retail Outlet Stores. The account balances (in thousands) are for 2017. 1. Compute (a) the cost of goods purchased and (b) the cost of goods sold. 2....
-
Via Gelato is a popular neighborhood gelato shop. The company has provided the following data concerning its operations: While gelato is sold by the cone or cup, the shop measures its activity in...
-
Develop an algorithm, in the form of a Raptor flowchart, to compute and display the amount of federal income tax to be paid when provided with the income as input. The Canadian Federal income tax...
-
Balance the following equations. (a) Zn(s) + VO+ (aq) Zn+ (aq) + V+ (aq) (b) Zn(s) + VO3(aq) V2+ (aq) + Zn+ (aq) (c) Zn(s) + CIO- (aq) Zn(OH)(s) + Cl(aq) (d) CIO- (aq) + [Cr(OH)4](aq) Cl(aq) +...
-
Use E values to predict which of the following metals, if coated on nickel, will provide cathodic protection against corrosion to nickel. (a) Cu (b) Mg (c) Zn (d) Cr
-
Vanderheiden Press Inc. and the Herrenhouse Publishing Company had the following balance sheets as of December 31, 2007 (thousands of dollars): Earnings before interest and taxes for both firms are...
-
The revenue (in dollars) from the sale of x units of a certain product can be described by R(x) = 100x-0.01x2. Find the instantaneous rate of change of the marginal revenue. R"(x)=2 Need Help? Read...
-
Clifford, Inc., has a target debt-equity ratio of .85. Its WACC is 8.1 percent, and the tax rate is 21 percent. a. If the company's cost of equity is 12 percent, what is its pretax cost of debt? (Do...
-
(1 point) A very large tank initially contains 100L of pure water. Starting at time t = 0 a solution with a salt concentration of 0.6kg/L is added at a rate of 5L/min. The solution is kept thoroughly...
-
(m) The waveform of signal A is shown in figure (1). Draw the output waveform of Z after execution of the following two statements : 08-0 Z
-
The Continental Bank advertises capital savings at 6.3% compounded annually while TD Canada Trust offers premium savings at 6.23% compounded quarterly. Suppose you have $3000 to invest for two years....
-
Norman Cross was the independent auditor for Home-Stakes Production Company, a company that offered investors interests in oil and gas drilling programs. The programs offered investors both income...
-
If a test has high reliability. O the test measures what the authors of the test claim it measures O people who take the same test twice get approximately the same scores both times O scores on the...
-
Draw an SM chart for the square root circuit of Problem 4.14. Data from Problem 4.14. This problem involves the design of a circuit that finds the square root of an 8-bit unsigned binary number N...
-
Draw an SM chart for the binary multiplier of Problem 4.22. Data from Problem 4.22. Design a multiplier that will multiply two 16-bit signed binary integers to give a 32-bit product. Negative numbers...
-
Design a binary-to-BCD converter that converts a 10-bit binary number to a 3-digit BCD number. Assume that the binary number is 999. Initially the binary number is placed in register B. When an St...
-
Demonstrates limited knowledge of health problem including social determinants and behavioral determinants. Also discuss what is the difference between them.
-
Sunnyvale Computer Company sells a line of computers that carry a six-month warranty. Customers are offered the opportunity to buy a two-year extended warranty for an additional charge. During 2016,...
-
Consider the following data to answer question 12 below. The figures are in millions of dollars. Balance sheet Cash and short-term investments 7000 Current liabilities 20000 Other current assets...

Study smarter with the SolutionInn App


