Methylacetamide, CH 3 CONHCH 3 , is a small molecule with an amide link (CONH), the group
Question:
Methylacetamide, CH3CONHCH3, is a small molecule with an amide link (CO—NH), the group that binds one amino acid to another in proteins.
(a) Is this molecule polar?
(b) Where do you expect the positive and negative charges to lie in this molecule? Does the electrostatic potential surface confirm your predictions?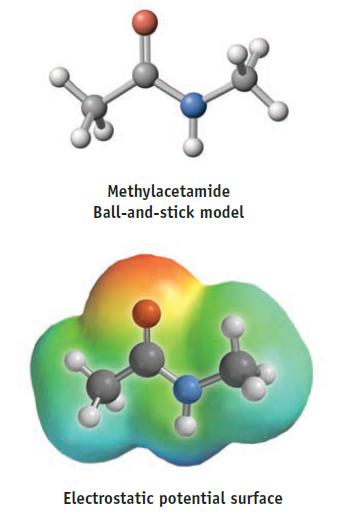
Transcribed Image Text:
Methylacetamide Ball-and-stick model Electrostatic potential surface
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
a Methylacetamide CH3CONHCH3 is a polar molecule The presence of the carbonyl group CO and the a...View the full answer

Answered By

Mary Boke
As an online tutor with over seven years of experience and a PhD in Education, I have had the opportunity to work with a wide range of students from diverse backgrounds. My experience in education has allowed me to develop a deep understanding of how students learn and the various approaches that can be used to facilitate their learning. I believe in creating a positive and inclusive learning environment that encourages students to ask questions and engage with the material. I work closely with my students to understand their individual learning styles, strengths, and challenges to tailor my approach accordingly. I also place a strong emphasis on building strong relationships with my students, which fosters trust and creates a supportive learning environment. Overall, my goal as an online tutor is to help students achieve their academic goals and develop a lifelong love of learning. I believe that education is a transformative experience that has the power to change lives, and I am committed to helping my students realize their full potential.
5.00+
4+ Reviews
22+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Accounting software that is designed for home use is often referred to as personal accounting software. This type of software is used mostly in managing household budgets and expenses. Some personal...
-
Acrylonitrile, C 3 H 3 N, is the building block of the synthetic fiber Orlon. (a) Give the approximate values of angles 1, 2, and 3. (b) Which is the shorter carboncarbon bond? (c) Which is the...
-
Cherokee Company had an accounts receivable balance of $16,000 on December 31, 2013. Projected sales for the first three months of 2014 are: January $120,000 February 130,000 March 100,000 All sales...
-
Describe one application, tool, and developer characteristic that should be assessed when evaluating whether or not a given application should be user developed, including what you see as the...
-
Why HR managers are getting alienated from grass root employees? How can that be reversed?
-
What is the significance of the 0/100 rule and the 50/50 rule and why would each be used?
-
Identify where the cash flow effect of each of the following transactions is reported in the statement of cash flows: operating, investing, or financing section. State the direction of each change....
-
Lindt, a Swiss chocolate producer, has sold fine chocolates in the United States. To win sales against other competitors, Lindt offers its overseas buyers attractive payment terms and allows them to...
-
A paper published in the research journal Science in 2007 (S. Vallina and R. Simo, Science, Vol. 315, p. 506, January 26, 2007) reported studies of dimethylsulfide (DMS), an important greenhouse gas...
-
Guanine is present in both DNA and RNA. (a) What is the most polar bond in the molecule? (b) What is the NC=N angle in the 6-member ring? (c) What is the NC=N angle in the 5-member ring? (d) What is...
-
In Problem find i (the rate per period) and n (the number of periods) for each loan at the given annual rate. Quarterly payments of $975 are made for 10 years to repay a loan at 9.9% compounded...
-
Find the slope-intercept form of the line with the given slope and y-intercept. 9 3 Slope = 2 y-intercept = 5 What is the equation of the line? (Simplify your answer. Type your answer in...
-
Discuss how terminal and instrumental values impact ethical workplace behavior
-
From the long 19th century to the short 20th century (i) Explains the relevance of extractive and metallurgical activities for the promotion of international industrial development at the end of the...
-
You invested $4500 four years ago. Today you have a balance of $6200. Calculate the effective annual rate of interest (as a %, to 2 decimal places) you earned on the investment. (Solve using excel...
-
What's in a wage? From pizza shop owners to house cleaners, voices on raising Maryland hourly minimum to $15. So, does everyone benefit from an increase in the minimum wage? Who could be the winners?...
-
You have heard that if you leave your money in mutual funds for a longer period of time, you will see a greater return. So you would like to compare the 3-year and 5-year returns of a random sample...
-
The power company must generate 100 kW in order to supply an industrial load with 94 kW through a transmission line with 0.09 resistance. If the load power factor is 0.83 lagging, find the...
-
Why must an acceptable wave function be single valued?
-
Why must the first derivative of an acceptable wave function be continuous?
-
Why must a quantum mechanical operator AË satisfy the relation l(x)[(x)] dx = f(x)[(x)]* dx?
-
Find an example of a company's aggregate planning strategy. Analyze the company's aggregate planning strategy and then provide the following. Background on the company processes Outline their...
-
Write a document in which: Provide examples of the different types of relationships between supplier and buyer. Discuss the advantages and disadvantages of each relationship. Answer the question: In...
-
Enter the forum and write a post discussing the role of government and compliance with foreign laws regarding buyer and supplier agreements. Your response must be substantial and analyze the topic...

Study smarter with the SolutionInn App


