Phenol (C 6 H 5 OH), commonly called carbolic acid, is a weak organic acid. If you
Question:
Phenol (C6H5OH), commonly called carbolic acid, is a weak organic acid.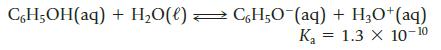
If you dissolve 0.195 g of the acid in enough water to make 125 mL of solution, what is the equilibrium hydronium ion concentration? What is the pH of the solution?
Transcribed Image Text:
CH₂OH(aq)+ H₂O(l) → CH₂O¯(aq) + H₂0+ (aq) K₂ 1.3 X 10-10 =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To find the equilibrium hydronium ion concentration and the pH of the solution we can use the inform...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Phenol, C 6 H 5 OH, is a weak organic acid. Suppose 0.515 g of the compound is dissolved in enough water to make 125 mL of solution. The resulting solution is titrated with 0.123 M NaOH. (a) What is...
-
You make a solution by dissolving 0.0010 mol of HCl in enough water to make 1.0 L of solution. a. Write the chemical equation for the reaction of HCl(aq) and water. b. Without performing...
-
Assume you dissolve 0.235 g of the weak acid benzoic acid, C 6 H 5 CO 2 H, in enough water to make 1.00 10 2 mL of solution and then titrate the solution with 0.108 M NaOH. (a) What was the pH of...
-
FIGURE P15.62 is a top view of an object of mass m connected between two stretched rubber bands of length L. The object rests on a frictionless surface. At equilibrium, the tension in each rubber...
-
Under what conditions could it be considered acceptable to hire a relative of an employee or executive in a public organization, such as in federal government, or a publicly owned business, such as...
-
Why are supporters of the efficient markets hypothesis unconvinced that differences between the theoretical and actual behavior of financial markets actually invalidate the hypothesis?
-
Both the suction and discharge piping for the pump shown in Fig. P12.22 consist of 4-in. I.D. 40-ft-long plastic pipe. Find the volume flow rate of \(60{ }^{\circ} \mathrm{F}\) water through the...
-
Although the BackPack Company has always used a level aggregate plan, Jill is interested in evaluating chase aggregate plans also. She has asked you to calculate how many hires and fires would be...
-
Review the major theoretical models , including the neuroscience, evolutionary, behavior genetics, psychodynamic, behavioral, cognitive, and social-cultural theoretical models. Think about the major...
-
What are the equilibrium concentrations of H 3 O + , CN , and HCN in a 0.025 M solution of HCN? What is the pH of the solution?
-
A 15-L flask at 300 K contains 6.44 g of a mixture of NO 2 and N 2 O 4 in equilibrium. What is the total pressure in the flask? (K p for 2 NO 2 (g) N 2 O 4 (g) is 7.1.)
-
Consider a single heat source located at the origin with temperature (a) Calculate the heat flux across the surface as shown in the figure. (b) Repeat the calculation in part (a) using the...
-
Because the decay products in the above fission reaction are neutron rich, they will likely decay by what process? A. Alpha decay B. Beta decay C. Gamma decay The uranium isotope \({ }^{235}...
-
a. Is there a stable \({ }_{3}^{30} \mathrm{Li}\) nucleus? Explain how you made your determination. b. Is there a stable \({ }_{92}^{184} \mathrm{U}\) nucleus? Explain how you made your determination.
-
What is the daughter nucleus of the decay? The Curiosity rover sent to explore the surface of Mars has an electric generator powered by heat from the radioactive decay of \({ }^{238} \mathrm{Pu}\), a...
-
How many protons and how many neutrons are in (a) \({ }^{3} \mathrm{He}\), (b) \({ }^{20} \mathrm{Ne}\), (c) \({ }^{60} \mathrm{Co}\), and (d) \({ }^{226} \mathrm{Ra}\) ?
-
It The wavelengths of light emitted by a firefly span the visible spectrum but have maximum intensity near \(550 \mathrm{~nm}\). A typical flash lasts for \(100 \mathrm{~ms}\) and has a power of...
-
Prince Rupert Fly n Fish Inc. purchases one small plane in its first year of business for $70,000. In year 2, it purchases another plane for $90,000. Find the UCC at the end of year 3 if the CCA rate...
-
Why is homeostasis defined as the "relative constancy of the internal environments? Does negative feedback or positive feedback tend to promote homeostasis?
-
Solve the equations in Prob. A18 using the Gauss elimination method. Data From Prob. A-18 4x 1 + x 2 + x 3 = -1, -5x 1 + 4x 2 + 3x 3 = 4, x 1 - 2x 2 + x 3 = 2 Using the matrix equation x = A -1 C.
-
Solve the equations x 1 + 2x 2 - 2x 3 = 5, x 1 - x 2 + x 3 = -1, x 1 - x 2 - x 3 = 1 Using the matrix equation x = A -1 C.
-
Solve the equations in Prob. A20 using the Gauss elimination method. Data From Prob. A-20 x 1 + 2x 2 - 2x 3 = 5, x 1 - x 2 + x 3 = -1, x 1 - x 2 - x 3 = 1 using the matrix equation x = A -1 C.
-
Mike's Auto Parts and Repairs Service's income statement data for the year ended August 31, 2018, follow. (Click the icon to view the data.) Assume the ending inventory was accidentally overstated by...
-
KFC Beef, a fast-food company selling roasted beef in outlets through the country, went public in 1993. In the year prior to going public, it had revenues of $40 million, on which it reported EBIT of...
-
What are the key differences between competitive, non-competitive, and uncompetitive enzyme inhibition? How are these concepts applied in the development of enzyme-based therapeutics ?

Study smarter with the SolutionInn App


