The backbone of both DNA and RNA consists of alternating deoxyribose and phophate groups . Figure 8.14
Question:
The backbone of both DNA and RNA consists of alternating deoxyribose and phophate groups .
Figure 8.14
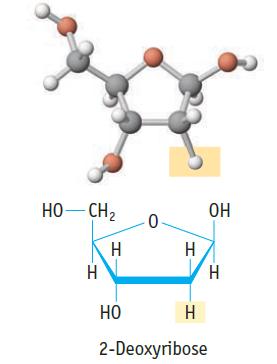
(a) Rank the types of bonds (such as C–C, C–O) in 2-deoxyribose in terms of increasing polarity.
(b) What are the expected C–O–C, O–C–C, and C–C–C bond angles in the C4O ring?
Transcribed Image Text:
HO-CH2 H I НО -0- H H 2-Deoxyribose ОН H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a CO CC CO bond is more polar than CC bond becau...View the full answer

Answered By

Shubhradeep Maity
I am an experienced and talented freelance writer passionate about creating high-quality content. I have over five years of experience working in the field and have collaborated with several renowned companies and clients in the SaaS industry.
At Herman LLC, an online collective of writers, I generated 1,000+ views on my content and created journal content for 100+ clients on finance topics. My efforts led to a 60% increase in customer engagement for finance clients through revamping website pages and email interaction.
Previously, at Gerhold, a data management platform using blockchain, I wrote and published over 50 articles on topics such as Business Finance, Scalability, and Financial Security. I managed four writing projects concurrently and increased the average salary per page from $4 to $7 in three months.
In my previous role at Bernier, I created content for 40+ clients within the finance industry, increasing sales by up to 40%.
I am an accomplished writer with a track record of delivering high-quality content on time and within budget. I am dedicated to helping my clients achieve their goals and providing exceptional results.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
A weekly payroll register shows that David Wu had total wages of $397.37. He had deductions of $30.40 for FICA tax, $41.25 for federal income tax, and $17.20 for medical insurance. Compute Wu's net...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
Which will undergo the greater rate of cooling: a red-hot poker in a warm oven or a red-hot poker in a cold room (or do both cool at the same rate)?
-
Which do you think are the two or three most important financial ratios? Why?
-
Identify some constraints in marketing to a traditional Muslim society. Use some of the examples in the chapter.
-
The following data refer to a compound impulse turbine having two rows of moving blades and one row of fixed blades in between them. Nozzle angle \(=15^{\circ}\), Exit velocity of steam from the...
-
What type of demand exists for pharmaceutical and medical device products? Discuss the roles doctors play in the business buying process for medical equipment used in a hospital and why companies...
-
Give your opinion/response to the below- Lukis Anderson arrested December 2012, for murder of Raveesh Kumra but actually it was more of framed by his own DNA. His DNA was found under the finger nail...
-
White phosphorus exists as P 4 molecules with phosphorus atoms at the corners of a tetrahedron. (a) Elemental phosphorus reacts with Cl 2 to form PCl 3 . Write a balanced chemical equation for this...
-
1,2-Dichloroethylene can be synthesized by adding Cl 2 to the carboncarbon triple bond of acetylene. Using bond dissociation enthalpies, estimate the enthalpy change for this reaction in the gas...
-
Suppose the tax rate on interest income is 40%, and the average tax rate on capital gains and dividend income is 15%. How high must the marginal corporate tax rate be to offer a tax advantage?
-
Sales on a bill-and-hold basis involve selling products but not delivering those products until a later date. 42 Sales on this basis have the effect of accelerating the recognition of those sales...
-
Given the following financial statement data, calculate the net operating cycle for this company. The net operating cycle of this company is closest to: A. 3.8 days. B. 24.3 days. C. 51.7 days. In...
-
The acquisition of Sky Systems by Modern Auto and the acquisition of Sky Systems by HiFly, respectively, would be examples of a: A. vertical merger and a horizontal merger. B. conglomerate merger and...
-
Which company had the highest number of days of receivables for the year 20X1? A. Company A. B. Company B. C. Company C. Mary Gonzales is evaluating companies in the office supply industry and has...
-
Given the following financial statement data, calculate the operating cycle for this company. The operating cycle for this company is closest to: A. 42.0 days. B. 47.9 days. C. 78.5 days. In millions...
-
You have completed an audit of activities within the purchasing department of your company. The department employs 30 buyers, seven supervisors, a manager, and clerical personnel. Purchases total...
-
An example of prescriptive analytics is when an action is recommended based on previously observed actions. For example, an analysis might help determine procedures to follow when new accounts are...
-
Solve Prob. 1061 using the maximum distortion energy theory Data from Problem: 10-61. The yield stress for a zirconium-magnesium alloy is Y = 15.3 ksi. If a machine part is made of this material and...
-
If a machine part is made of tool L2 steel and a critical point in the material is subjected to in-plane principal stresses 1 and 2 = -0.5 1 , determine the magnitude of 1 in ksi that will cause...
-
Solve Prob. 1063 using the maximum distortion energy theory. Problem: 10-63 If a machine part is made of tool L2 steel and a critical point in the material is subjected to in-plane principal stresses...
-
Consider a spherical bacterium, with radius 1.4 m , falling in water at 20 C. Find the terminal speed of the spherical bacterium in meters per second, ignoring the buoyant force on the bacterium and...
-
Calculate C in the units of d 2 /AU 3 for each orbital body listed in this table. Use the formula T 2 = Ca 3 and the proper number of significant digits in your value for C. The first one has been...
-
Ursula, Wendy, Xavier, Yvonne, and Zack, a group of first year law students decided to throw a party in their house to celebrate passing their exams. The following events took place: (a) Wendy was in...

Study smarter with the SolutionInn App


