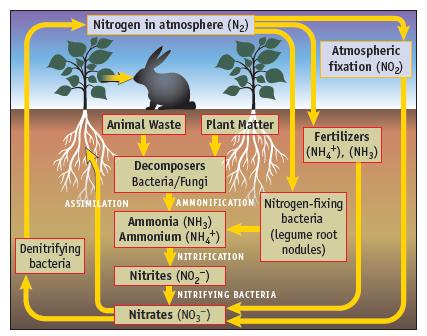
The nitrogen cycle (Figure 20.1) shows the oxidation of NH 4 + , first to NO 2
Question:
The nitrogen cycle (Figure 20.1) shows the oxidation of NH4+, first to NO2− and then the subsequent oxidation of NO2− to NO3−. Write balanced equations for each of these half-reactions (in acid solution).
Data given in Figure 20.1
Transcribed Image Text:
Nitrogen in atmosphere (N₂) Denitrifying bacteria Animal Waste Plant Matter ASSIMILATION Decomposers Bacteria/Fungi Ammonia (NH3) Ammonium (NH4+) AMMONIFICATION Nitrogen-fixing bacteria NITRIFICATION Nitrites (NO₂) Atmospheric fixation (NO₂) NITRIFYING BACTERIA Nitrates (NO3) Fertilizers (NH,*), (NH3) (legume root nodules)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The following are balanced equations for the oxidation of NH4 to NO2 and the oxidation of NO2 to NO3 ...View the full answer

Answered By

Sidharth Jain
My name is Sidharth. I completed engineering from National Institute of Technology Durgapur which is one of the top college in India. I am currently working as an Maths Faculty in one of the biggest IITJEE institute in India. Due to my passion in teaching and Maths, I came to this field. I've been teaching for almost 3 years.
Apart from it I also worked as an Expert Answerer on Chegg.com. I have many clients from USA to whom I teach online and help them in their assignments. I worked on many online classes on mymathlab and webassign. I guarantee for grade 'A'.
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Write balanced equations for each of the following reactions (some of these are analogous to reactions shown in the chapter). (a) Aluminum metal reacts with acids to form hydrogen gas. (b) Steam...
-
Write balanced equations for the reactions described in Table 18.13 for the production of Bi and Sb. Table 18.13 Element Nitrogen Phosphorus Electronegativity Source Method of Preparation 3.0 2.2 Air...
-
Write balanced equations for each of the following. a. Solid calcium fluoride is heated with sulfuric acid to give hydrogen fluoride vapor. b. Solid potassium chlorate is carefully heated to yield...
-
Can you give us a few Google AdWords Ad Extensions names that you know?
-
Integrated Technologies Inc. is considering the purchase of automated machinery that is expected to have a useful life of four years and no residual value. The average rate of return on the average...
-
A manufacturing company sells motorcycles with a five-year warranty against manufacturers defects. The manufacturer expects that 0% of the motorcycles sold will prove to be defective in the first...
-
A study found that eating a few pieces of chocolate each week can improve your cardiovascular health. Identify the two events described in the study. Do the results indicate that the events are...
-
Operating data for Gladow Corporation are presented below. InstructionsPrepare a schedule showing a vertical analysis for 2012 and 2011. 2012 2011 Sales $800,000 520,000 $600,000 Cost of goods sold...
-
Your client has contacted you because they can't add a product/service to their invoice. Why might this happen? They do not have permission to add products or services to invoices They turned off...
-
Refer to the figure below and Figure 20.25, which show the fraction of species in solution [alpha ()] as a function of pH. The following questions are in regard to the equilibria involved in an...
-
The refrigerating liquids in air conditioners and refrigerators are largely chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs). Among the latter family of compounds is the refrigerant...
-
Construct a table comparing the structure of arteries, veins and capillaries. Include both similarities and differences, and give reasons for the differences which you describe.
-
In what ways can strategic foresight methodologies, such as horizon scanning or trend analysis, inform strategic planning processes by identifying emerging opportunities, disruptive threats, and...
-
How can strategic innovation frameworks, such as design thinking or blue ocean strategy, inspire breakthrough innovations and create new market spaces by challenging conventional assumptions and...
-
Consider the following questions in connection with the view of Justice Brandeis in Ashwander v. TVA: 1. Under what circumstances has the Supreme Court asserted that, despite jurisdiction, a court...
-
IN ACFR, if government entity increases its legal debt limit, the legal debt margin also increases what it means?
-
Subject: Constitutional Law (Malaysia) Question: DISCUSS critically the constitutional impact and significance of the following decided cases with special emphasis on the freedom of speech and...
-
In Exercise 12.35, we described how the office equipment chain OfficeMax offers rebates on some products. The goal in that exercise was to estimate the total amount spent by customers who bought the...
-
Prepare a stock card using the following information A company is registered for GST which it pays quarterly, assume GST was last paid on the 30th of June 2019. It uses weighted average cost...
-
Derive the state transition table and D flip-flop input equations for a counter that counts from 1 to 6 (and back to 1 and continues).
-
Reduce the following state table to a minimum number of states. Present State Next State Output X = 1 X = 0 X= 1 B A D D G D
-
A Mealy sequential circuit is implemented using the circuit shown in Problem 1.26. Assume that if the input X changes, it changes at the same time as the falling edge of the clock. (a) Complete the...
-
Presented below is information related to Tamarisk Corporation at December 31, 2025, the end of its first year of operations. Sales revenue Cost of goods sold $536,000 360,000 Interest expense 31,600...
-
John purchases State of Oklahoma general-purpose bonds at a cost of $3,400 in 2018. He receives $210 interest on the bonds in 2018 2019, and 2020. In 2020, he sells the bonds for $3,800. How much...
-
Pesto Company paid $8 per share to acquire 80% of Sauce Company's 100,000 outstanding shares. At the acquisition, the fair value of Sauce's Total assets $1,000,000 and the fair value of Sauce's total...

Study smarter with the SolutionInn App


