The photo below shows elemental iodine dissolving in ethanol to give a solution. Is this a physical
Question:
The photo below shows elemental iodine dissolving in ethanol to give a solution. Is this a physical or chemical change? 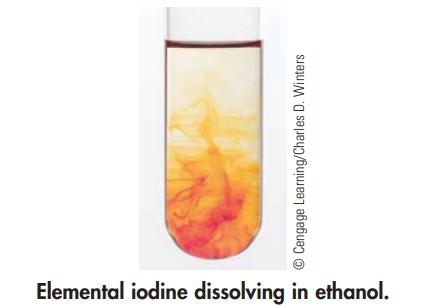
Transcribed Image Text:
Ⓒ Cengage Learning/Charles D. Winters Elemental iodine dissolving in ethanol.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
The process of elemental iodine dissolving in ethanol to form a solution is a physical change In a ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
What is the maturity value of a $5,579.00 loan borrowed at 3.79% compounded semi- annually for 4 years and 4 months? DO NOT ROUND INTERMEDIATE RESULTS. Report N accurate to at least 6 decimal places....
-
A barium mineral was dissolved in hydrochloric acid to give a solution of barium ion. An excess of potassium sulfate was added to 50.0 mL of the solution, and 1.128 g of barium sulfate precipitate...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
SG Company acquired 80% of Popsters Company on January 1, 2019, when the stockholders equity of Popsters consisted of: Ordinary shares, P100 par P500,000 Paid in capital in excess of par 400,000...
-
Determine the due date and the amount of interest due at maturity on the followingnotes: Date of Note Face Amount Term of Note Interest Rate a. June 2 b. August 30 c. October 1 d. March 6 e. May 20...
-
The comparative balance sheet of Coulson, Inc. at December 31, 20Y2 and 20Y1, is as follows: Dec. 31, 20Y2 Dec. 31, 20Y1 Assets Cash . . . . Accounts receivable (net)... . 300,600 704,400 918,600...
-
Below is a list of transactions completed by Recycled Fashion during 2024. Ignore GST. For each transaction, indicate (a) the section (i.e. operating, investing or financing) of the statement of cash...
-
The following is a partial list of the account balances, after adjustments, of the Silvoso Company on December 31, 2007: The following information is also available: 1. The company declared and paid...
-
If Ax+By+5z=C is an equation for the plane containing the point (0,0,1) and the line x-1=Y+2, z=72, 3 then A+B+C=
-
A copper-colored metal is found to conduct an electric current. Can you say with certainty that it is copper? Why or why not? Suggest additional information that could provide unequivocal...
-
Four balloons are each filled with a different gas, each having a different density: If the density of dry air is 1.12 g/L, which balloon or balloons float in air? helium, d = 0.164 g/L argon, d =...
-
Determine the polar moment of inertia and the polar radius of gyration of the shaded area shown with respect to point P.
-
(2) Example (d) in section 16.8 of the book shows the derivation of the flux density of a thin homogenously charged ring (radius R, charge Q) in the symmetry axis of the ring at distance x Q X 4...
-
how to make a XOR and mux in Hardware simulator(2.5 ). The HDL codes I will post in below then (If need make change code please show me too by screen short .) You can draw your design on paper and...
-
Evaluate the following limit. t t lim costi-2 sinj+k t lim costi-2 sinj+k
-
Melissa Cutt is thinking about buying some shares of EZLawn Equipment, at $36.44 per share. She expects the price of the stock to rise to $43.62 over the next 3 years. During that time she also...
-
The assumption behind most strategy models is that management is rational and can see the world objectively. Do you believe this is true? What can keep managers from being purely rational and...
-
Cal Lopez discovered significant fraud in the accounting records of a high profile client. The story has been broadcast on national airways. Lopez was unable to resolve his remaining concerns with...
-
Don Griffin worked as an accountant at a local accounting firm for five years after graduating from university. Recently, he opened his own accounting practice, which he operates as a corporation....
-
In Chapter 21, we will explore how nitriles can be converted into carboxylic acids. How would you use IR spectroscopy to monitor the progress of this reaction? R-CEN A nitrile ,* C. A carboxylic acid
-
1-Butyne was treated with sodium amide followed by ethyl iodide. An IR spectrum of the product was acquired and then compared with the spectrum of the starting alkyne. A signal at 2200 cm -1 was...
-
Each of the following compounds contains two carbonyl groups. Identify which carbonyl group will exhibit a signal at lower wave number. a. b. c. O:
-
How often should urinalysis be performed to screen for microalbuminuria in the management of a patient with type 2 diabetes?
-
Whats the role of underwriters and prospectuses in IPO? What is the key difference between IPO and SEO? Why SEO also has the underpricing phenomenon?
-
A firm with a cost of capital of 7% is considering two projects (they are not mutually exclusive), one expected to return 9% and a second expected to return 9.5%. What projects should it pursue?

Study smarter with the SolutionInn App


