Use data in Appendix L to calculate the enthalpy and free energy change for the reaction 2
Question:
Use data in Appendix L to calculate the enthalpy and free energy change for the reaction 2 NO(g) + O2(g) → 2 NO2(g). Is this reaction exothermic or endothermic? Is the reaction product- or reactant-favored at equilibrium?
Data given in Appendix L
Transcribed Image Text:
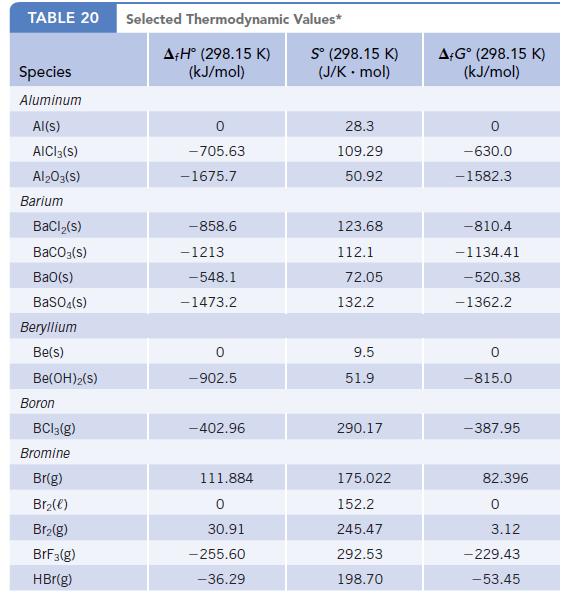
TABLE 20 Species Aluminum Al(s) AICI 3(S) Al2O3(S) Barium BaCl₂(s) BaCO3(s) BaO(s) BaSO4(s) Beryllium Be(s) Be(OH)2(S) Boron BC13(g) Bromine Br(g) Br₂(e) Br₂(g) BrF3(g) HBr(g) Selected Thermodynamic A+Hº (298.15 K) (kJ/mol) 0 -705.63 -1675.7 -858.6 -1213 -548.1 -1473.2 -902.5 -402.96 111.884 0 30.91 -255.60 -36.29 Values* Sº (298.15 K) (J/K . mol) 28.3 109.29 50.92 123.68 112.1 72.05 132.2 9.5 51.9 290.17 175.022 152.2 245.47 292.53 198.70 A+Gᵒ (298.15 K) (kJ/mol) -630.0 -1582.3 -810.4 -1134.41 -520.38 -1362.2 0 -815.0 -387.95 82.396 0 3.12 - 229.43 -53.45
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Enthalpy change To calculate the enthalpy change for the reaction we can use the following equation ...View the full answer

Answered By

ZIPPORAH KISIO LUNGI
I have worked on several other sites for more than five years, and I always handle clients work with due diligence and professionalism. Am versed with adequate experience in the fields mentioned above in which have delivered quality papers in research, thesis, essays, blog articles, and so forth.
I have gained extensive experience in assisting students to acquire top grades in biological, business and IT papers. Notwithstanding that, I have 7+ years of experience in corporate world software design and development.
5.00+
194+ Reviews
341+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Use data in Appendix L to calculate the enthalpy and free energy change for the reaction 2 NO 2 (g) N 2 O 4 (g) Is this reaction exothermic or endothermic? Is the reaction product- or...
-
The stratospheric ozone (O3) layer helps to protect us from harmful ultraviolet radiation. It does so by absorbing ultraviolet light and falling apart into an O2 molecule and an oxygen atom, a...
-
The free energy change for a reaction G is an extensive property. What is an extensive property? Surprisingly, one can calculate G from the cell potential for the reaction. This is surprising because...
-
During 2018, Susan incurred and paid the following expenses for Beth (her daughter), Ed (her father), and herself: Surgery for...
-
Most jurisdictions have two possible verdicts in criminal trials: guilty or not guilty. Scottish criminal trials have three possible verdicts: guilty, not proven, or not guilty. Explain the...
-
Find i(t) for t > 0 in the circuit of Fig. 16.43 . t = 0 10 60 ww |(t) 1 mF 40 36 V (+ 2.5 H (+1
-
Consider a single-mode laser emitting light described by the analytic signal \[ \mathbf{u}(t)=\exp (-j[2 \pi \bar{v} t-\theta(t)]) \] (a) Assuming that \(\Delta \theta=\theta\left(t_{2}...
-
Alton Newman, age 67, is married and files a joint return with his wife, Clair, age 65. Alton and Clair are both retired, and during 2016, they received Social Security benefits of $10,000. Both...
-
We are given the following information for Pettit Corporation. Sales (credit) Cash Inventory Current liabilities Asset turnover Current ratio Debt-to-assets ratio Receivables turnover $3,018,000...
-
The overall reaction involved in the industrial synthesis of nitric acid is NH 3 (g) + 2 O 2 (g) HNO 3 (aq) + H 2 O() Calculate r G for this reaction and its equilibrium constant at 25C.
-
A major use of hydrazine, N 2 H 4 , is in steam boilers in power plants. (a) The reaction of hydrazine with O 2 dissolved in water gives N 2 and water. Write a balanced equation for this reaction....
-
1. In this scenario, management was able to outline some specific characteristics that the resulting technology must have. Is this a good idea? On the one hand, it provides clear criteria against...
-
A muon is created 60 km above the surface of the earth. In the earths frame, the muon is traveling directly downward at 0.9999c. a. In the earths frame of reference, how much time elapses between the...
-
In an intergalactic competition, spaceship pilots compete to see who can cover the distance between two asteroids in the shortest time. The judges are at rest with respect to the two asteroids. From...
-
Geraldo is a newsman embedded with an Army infantry unit in Afghanistan. In an effort to give his network the best coverage of the war on terror, Geraldo draws a map in the sand indicating where his...
-
Robbery consists of (1) the taking (2) and carrying away (3) of the personal property (4) of another person (5) by violence or by putting the victim in fear (6) with intent to permanently deprive the...
-
Rape is generally defined as the taking of sexual intimacy with an unwilling person by force or without consent. Rape is usually intercourse; sexual assault is other sexual contact without consent. ...
-
Problem described some adjustments made by Alaska Airlines. The adjustments are lettered (a) through (f). Repeat the requirements for each adjustment as it would be made by the other party in the...
-
d. The characteristic equation of a control system is given by s+2s+8s+12s+20s+16+16=0. Determine the number of the roots of the equation which lie on the imaginary axis of s-plane
-
A typical human baby at birth is about 20 inches as measured from head to toe. An adult is much longer. What is the approximate ratio of the height of an adult female to the length of a baby at birth?
-
The solar system in perspective. The diameters of the Earth and the Sun are approximately 1.3 10 7 m and 1.4 10 9 m respectively, and the average SunEarth distance is 1.5 10 11 m. Consider a scale...
-
Estimate the number of times a students heart will beat during the 4 years she is a college student.
-
Calculate the price of a three-month European put option on a non-dividend-paying stock with a strike price of $50 when the current stock price is $50, the risk-free interest rate is 10% per annum,...
-
Danielle & Company Limited (DCL), based in Portland, has the following capital structure: debt is 41%, preferred stock is 28% and common stock is 31%. Their tax rate is 25% and investors expect...
-
List all the pairs of integers with a product of -24. Then find the pair whose sum is - 23.

Study smarter with the SolutionInn App


