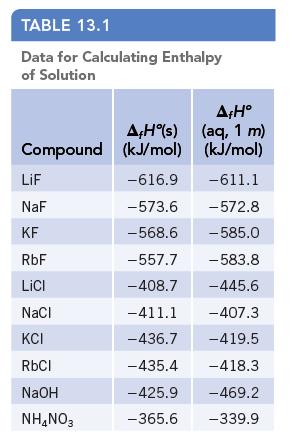
Use the data of Table 13.1 to calculate the enthalpy of solution of LiCl. Data given in
Question:
Use the data of Table 13.1 to calculate the enthalpy of solution of LiCl.
Data given in Table 13.1
Transcribed Image Text:
TABLE 13.1 Data for Calculating Enthalpy of Solution Compound LiF NaF KF RbF LICI NaCl KCI RbCl NaOH NH4NO3 AH(s) (kJ/mol) -616.9 -573.6 -568.6 -557.7 -408.7 -411.1 -436.7 -435.4 -425.9 -365.6 AfHº (aq, 1 m) (kJ/mol) -611.1 -572.8 - 585.0 -583.8 -445.6 - 407.3 -419.5 -418.3 -469.2 -339.9
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
To calculate the enthalpy of solution of LiCl using Table 131 ...View the full answer

Answered By

Morgan Njeri
Very Versatile especially in expressing Ideas in writings.
Passionate on my technical knowledge delivery.
Able to multitask and able to perform under pressure by handling multiple challenges that require time sensitive solution.
Writting articles and video editing.
Revise written materials to meet personal standards and satisfy clients demand.
Help Online Students with their course work.
4.90+
12+ Reviews
38+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Diseases of the present age are very different from diseases of the past. Discuss.
-
Use the data of Table 7.1 to show that the effective area of a rectangular or circular paraboloidal antenna is roughly equal to its geometrical area. Table 7.1 HPBW (degrees) D (dB) Antenna type...
-
Use the data of Exercise 19 to calculate a 95% CI for the difference between true average stopping distance for cars equipped with system 1 and cars equipped with system 2. Does the interval suggest...
-
In year 1, the prices of goods X, Y, and Z are $2, $4, and $6 per unit, respectively. In year 2, the prices of good X, Y, and Z are $3, $4, and $7, respectively. In year 2, twice as many units of...
-
The search committee for a new CEO is seriously considering Carly Fiorina. Based on the information in the case, debate the controversy: should we hire this woman who is an outsider, or look for...
-
The following balance sheet and income statement data were taken from the records of Steeler and Jones for the year ended December 31, 2018: Prepare the operating section of the statement of cash...
-
With reference to the preceding exercise, change the equation obtained in part (a) to the form \(\widehat{y}=a \cdot e^{-c x}\), and use the result to rework part (b).
-
Blanka Transport, Inc. After several years of driving long-haul trucks, Joe Blanka founded his own trucking company, Blanka Transport Inc. (BTI), which specialized in less-than-carload shipments in...
-
Discuss the role of real-time communication protocols in RTOS-based systems. How do these protocols ensure real-time data exchange while maintaining system stability and predictability ?
-
Which pairs of liquids will be miscible? (a) H 2 O and CH 3 CH 2 CH 2 CH 3 (b) C 6 H 6 (benzene) and CCl 4 (c) H 2 O and CH 3 CO 2 H
-
Silver ion has an average concentration of 28 ppb (parts per billion) in U.S. water supplies. (a) What is the molality of the silver ion? (b) If you wanted 1.0 10 2 g of silver and could recover it...
-
In Problems 3360, find the exact value of each expression. sin- [cos(-77)] 6
-
Siemens is a German conglomerate that has been in business since 1847 with its three divisions of Energy, Health Care, and Industry. Siemens has 428,200 employees and operates in 190 countries,...
-
For the past five years, Congress has been considering a bill that would require commercial truckers to install electronic recorders, often called "black boxes" on all of their trucks. Currently,...
-
Following a 12-day trial in 2012, Walter Cardin, a safety manager for the Shaw Group, was convicted of eight counts of fraud against the United States, for falsifying injury reports for his company's...
-
In 2007, Eleazar Torres-Gomez fell into an industrial dryer at the Cintas plant where he worked. He was killed before anyone even noticed that he had fallen into the dryer from the moving conveyor...
-
The stock market of the late 1990 s and early 2000 s represented a period of irrational exuberance. Investors invested as they never had, egged on by analysts who could say no evil of the companies...
-
Use the financial statements for Fairplay Inc. provided in E12-5 to respond to the following: a. Prepare trend (horizontal analysis) balance sheets and income statements for 2016, 2017, and 2018. Use...
-
In the synthesis of the keto acid just given, the dicarboxylic acid decarboxylates in a specific way; it gives Explain. HO rather than HO
-
The beam supports the distributed load shown. Determine the resultant internal loadings acting on the cross section at point C. Assume the reactions at the supports A and B are vertical. 4 kN/m A...
-
The beam supports the distributed load shown. Determine the resultant internal loadings acting on the cross section at point D. Assume the reactions at the supports A and B are vertical. 4 kN/m -1.5...
-
Determine the resultant internal loadings acting on the cross sections at points D and E of the frame. 4 ft 75 Ib/ft LA B. 2 ft 2 ft 1 ft 1 ft 30 1 ft 150 lb
-
R1 Solve for the equivalent circuit resistance and the currents through R2 and R3 (Ans: 0.946mA. IR3 = 0.473mA) Req = 6.34k. IR2 w 2k 9v 24 R2 4k R4 1.67k R3 8k
-
1. Utada is riding on a carnival swing ride. The top spins around causing all the people on the swings to swing outward as shown below in the nice sketch on the left. The diagram on the right is for...
-
You are casting high carbon steel and need a 50:1 atomic ratio of Fe:C (fifty times as many Fe atoms as C atoms) in the powder you will melt and cast. If you use 2.3 kg (2300 g) of Fe powder, what is...

Study smarter with the SolutionInn App


