Which of the following compounds produces acetic acid when treated with an oxidizing agent such as KMnO
Question:
Which of the following compounds produces acetic acid when treated with an oxidizing agent such as KMnO4?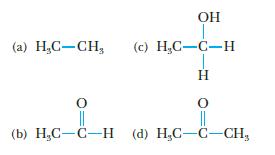
Transcribed Image Text:
(c) H.C-C-Н "HO-O-O'H (P) H-D-DH (9) || 0 H || HO 0 "HO-O'H (2)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The compound that produces acetic acid CH3COOH when treated with an oxidizing ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
Consider a unity-feedback control system whose feedforward plant is given by 10 s(s + 2)(s +8) G(s): = Design a lead-lag compensator such that the dominant closed-loop poles are lo- cated at s= -2j23...
-
Nitric acid is a strong oxidizing agent. State which of the following species is least likely to be produced when nitric acid reacts with a strong reducing agent such as zinc metal, and explain why:...
-
You make a one-time investment of $400 and leave it for 5 years, earning an annual interest rate of 7%. How much interest will you have earned after 5 years? If you leave the entire amount in your...
-
Combes Furniture Company manufactures furniture. Combes uses a job order cost system. Balances on November 1 from the materials ledger are as follows: Fabric ..... $ 33,500 Polyester filling .. 8,100...
-
Jose Corporation has $4,000,000 of 9.5 percent, 25-year bonds dated March 1, 2010, with interest payable on February 28 and August 31. The companys fiscal year end is February 28. It uses the...
-
Use frequency distribution formulas to approximate the sample mean and the sample standard deviation of the data set in Exercise 1. Data from Exercise 1 The data set represents the numbers of minutes...
-
A hot fluid passes through circular channels of a cast iron platen (A) of thickness LA = 30 mm which is in poor contact with the cover plates (B) of thickness L B = 7.5 mm. The channels are of...
-
Dr. Mars has a mass of 75 kg and weighs 735 N or 165 lbs on the Earth. a) What is his weight in N and lbs on Jupiter where the acceleration due to gravity is 24.8 m/s2? b) If he weighs 63.1 lbs on...
-
Nylon-6 is a polyamide formed by polymerizing H 2 NCH 2 CH 2 CH 2 CH 2 CH 2 CO 2 H. Write an equation for this reaction.
-
Review the opening photograph about chocolate (which shows the structure of an active ingredient, the obromine) and then answer the following questions. (a) How do the obromine and caffeine differ...
-
Gasoline is piped underground from refineries to major users. The flow rate is 3.00 10-2 m3/s (about 500 gal/min), the viscosity of gasoline is 1.00 10 -3 (N/m 2 ) s, and its density is 680 kg/m3....
-
Discuss two (2) ways in which organisational policies and procedures may govern or inform the preparation and implementation of budgets and financial plans bsbfin501
-
Management has written a policy prohibiting employees from accepting any gifts valued above $50. Which component of the system of internal controls does this statement provide evidence of?
-
Describe any funding provided for capital expenditure. What is the purpose or value of the item(s) you identified? Some of the funded provide for capital expenditure for the program come from Local...
-
COGS and Inventory Valuation. 1. COGS is an acronym, what do the letters stand for? 2. There is a formula for calculating COGS.. list the formula. Ex.. COGS = Then, define each variable in the COGS...
-
To account for an insufficient funds check from a customer, you must have a service item for the bounced check. What account should you select for the income account when setting up the bounced check...
-
A popularly held belief about university professors is that they dont work very hard and that the higher their rank, the less work they do. A statistics student decided to determine whether the...
-
a) Calculate the goodwill that was paid by Major Ltd on the acquisition of Minor Ltd. [10 marks] b) Prepare the consolidated statement of financial position for Major Ltd at 31 July 20X8. [30 marks]...
-
A horse galloped a mile in 2 min 35 s. What was its average speed in km/h?
-
Figure P.4.95 is a plot of n I and n R versus λ for a common metal. Identify the metal by comparing its characteristics with those considered in the chapter and discuss its optical...
-
Convert 0.296 cm 3 /s to m 3 /s.
-
Duela Dent is single and had $190,400 in taxable income. Calculate her income taxes. Taxable Income Tax Rate 10% $ 0- 9,525 9,525- 38,700 12 38,700- 82,500 22 157,500 200,000 82,500-157,500...
-
Select a company you wish to analyze and evaluate from the "undercover employee" perspective. Troy mill wood products https://www.troymill.com/about/ You may or may not like the organization; that...
-
Final draft: Preparation of a sales presentation. Students will develop a sales pitch, an order development process, customer interaction, order delivery and closing documents to finalize the sale. ...

Study smarter with the SolutionInn App


