You titrate 25.0 mL of 0.10 M NH 3 with 0.10 M HCl. (a) What is the
Question:
You titrate 25.0 mL of 0.10 M NH3 with 0.10 M HCl.
(a) What is the pH of the NH3 solution before the titration begins?
(b) What is the pH at the equivalence point?
(c) What is the pH at the halfway point of the titration?
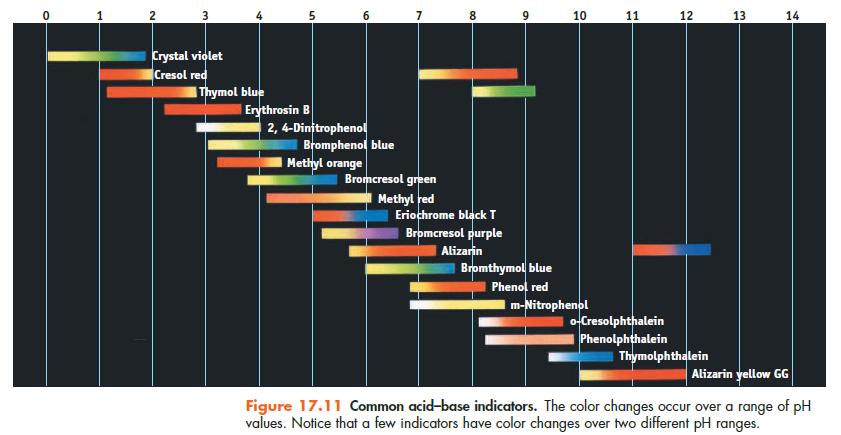
(d) What indicator in Figure 17.11 could be used to detect the equivalence point?
(e) Calculate the pH of the solution after adding 5.00, 15.0, 20.0, 22.0, and 30.0 mL of the acid. Combine this information with that in parts (a)–(c) and plot the titration curve.
Data given in Figure 17.11
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:





