Azomethane, CH 3 NNCH 3 , is not a stable compound, and once generated, it decomposes. The
Question:
Azomethane, CH3NNCH3, is not a stable compound, and once generated, it decomposes. The rate of decomposition was measured by monitoring the partial pressure of azomethane, in torr:
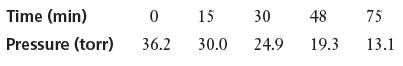
Plot the data and determine the instantaneous rate of decomposition of azomethane at t = 20 min.
Transcribed Image Text:
Time (min) Pressure (torr) 0 36.2 15 30 30.0 24.9 48 19.3 75 13.1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Estimating th...View the full answer

Answered By

Keziah Thiga
I am a self motivated financial professional knowledgeable in; preparation of financial reports, reconciling and managing accounts, maintaining cash flows, budgets, among other financial reports. I possess strong analytical skills with high attention to detail and accuracy. I am able to act quickly and effectively when dealing with challenging situations. I have the ability to form positive relationships with colleagues and I believe that team work is great key to performance. I always deliver quality, detailed, original (0% plagirism), well-researched and critically analyzed papers.
4.90+
1504+ Reviews
2897+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
There are two significant pieces of legislation that impact maritime security in the United States. These include the Maritime Transportation Security Act 2002 and the S.A.F.E. Port Act of 2006....
-
At 400 K, the rate of decomposition of a gaseous compound initially at a pressure of 12.6 kPa was 9.71 Pa S-1 when 10.0 per cent had reacted and 7.67 Pa S-I when 20.0 per cent had reacted. Determine...
-
The rate of the decomposition of hydrogen peroxide, H 2 O 2 , depends on the concentration of iodide ion present. The rate of decomposition was measured at constant temperature and pressure for...
-
(a) Find the Maclaurin series for the function f(x)= ln(1+x) and hence that for In(1+x) (b) By keeping the first four terms in the Maclaurin series for In(1+x) integrate the function In(1+x) from x =...
-
Management for Marlowe Manufacturing Company decided in 2010 to discontinue one of its unsuccessful product lines. (The product line does not meet the definition of a business component.) The planned...
-
1. How much cash is paid to acquire merchandise inventory during year 2011? 2. How much cash is paid for operating expenses during year2011? ORWELL, INC. Balance Sheets December 31,2011 2011 2010 2...
-
Refer to the information in QS 19-16. The company sells its product for $50 per unit. Due to new regulations, the company must now incur $2 per unit of hazardous waste disposal costs and $8,500 per...
-
Selected information from the payroll register of Anderson's Dairy for the week ended July 7, 20--, is shown below. The SUTA tax rate is 5.4%, and the FUTA tax rate is 0.8%, both on the first $7,000...
-
List and describe/discuss the five advantages of intermediate sanctions over traditional probation or incarceration.
-
In a method of initial rates experiment, is the measured rate an average rate or an instantaneous rate? Explain.
-
A gas, AB, decomposes and the volume of B 2 produced is measured as a function of time. The data obtained are as follows: What is the average rate of production of B 2 for the first 8.3 min? For the...
-
Earl Massey, director of marketing, wants to reduce the selling price of his company's products by 15% to increase market share. He says, "I know this will reduce our gross profit rate, but the...
-
A square loop with sides 1 2 cm by 1 2 cm has 2 5 turns of wire and is in a magnetic field of strength 4 . 2 x 1 0 ^ - 2 T with its axis perpendicular to the direction of the field. if it is rotated...
-
How do post-transcriptional and post-translational regulatory mechanisms, such as microRNA-mediated gene silencing and protein modification, contribute to the fine-tuning of developmental processes,...
-
Grandin Inc. is evaluating its dividend policy. It has a capital budget of $625,000, and it wants to maintain a target capital structure of 60% debt and 40% equity. The company forecasts a net income...
-
recording a lon term lease as an operating lease as opposed to a capital lease what will cause for a lease are the debt to equity will be higher than total asset turnover? Explain.
-
Consider a reaction that has a negative AH and a positive AS. Which of the following statements is true? This reaction will be nonspontaneous only at high temperatures. This reaction will be...
-
Ultra-Home Corporation provides the following information related to its defined-benefit pension plan for 2014. Pension liability (January 1) .............$ 500,000 Accumulated benefit obligation...
-
What can you do to reduce hunger where you live? To reduce hunger globally?
-
Identify the reagents necessary to make each of the following amino acids via the amidomalonate synthesis. (a) Isoleucine (b) Alanine (c) Valine
-
Each of the following carboxylic acids was treated with bromine and PBr3 followed by water, and the resulting α-haloacid was then treated with excess ammonia. In each case, draw and...
-
Each of the following aldehydes was converted into an -amino nitrile followed by hydrolysis to yield an amino acid. In each case, draw and name the amino acid that was produced. (a) Acetaldehyde (b)...
-
You are reviewing a corporation's financial statements and observe that the income tax expense has two components a current portion and a deferred portion. Explain which component appears on the...
-
A company receives a $900 utility bill for the current month but does not plan to pay the bill until early next month. Record the receipt of the utility billusing (a) accrual-basis accounting and (b)...
-
Define two cost accounting methods. Describe typical uses for each and include examples. Discuss which of the various cost accounting methods you think are most appropriate for measuring the costs of...

Study smarter with the SolutionInn App


