Calculate the enthalpy change when 245 g of ice melts. Strategy The H fus value in Table
Question:
Calculate the enthalpy change when 245 g of ice melts.
Strategy The ΔHfus value in Table 9.3 is in J/mol, so the amount of ice must be converted into moles. Multiplying the number of moles by ΔHfus will provide the desired quantity.
Table 9.3
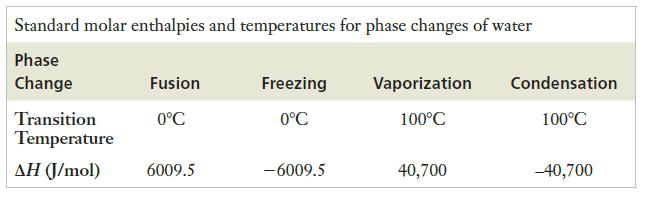
Transcribed Image Text:
Standard molar enthalpies and temperatures for phase changes of water Phase Change Transition Temperature AH (J/mol) Fusion 0°C 6009.5 Freezing 0°C -6009.5 Vaporization 100°C 40,700 Condensation 100°C -40,700
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Analyze Your Answer The enthalpy of fusion is constant at 6 kJmol so the enthalpy change depends on ...View the full answer

Answered By

Alexandra Scotg
My first teaching job came in 2014- I worked at a further education college, teaching people from ages 16 onwards. Then in 2017- after having my daughter, I needed a flexible job. I started my online teaching journey and 3 years later I still love it!
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
The enthalpy change when liquid methanol, CH3OH, vaporizes at 25oC is 38.0 kJ/mol. What is the entropy change when 1.00 mol of vapor in equilibrium with liquid condenses to liquid at 25oC? The...
-
We use the formula PV = FV (1/1+R)^N to calculate Present Value. The formula is used to figure out how much a future sum is worth today, given that there is always a discount rate. In class, we...
-
When 15.3 g of sodium nitrate, NaNO3, was dissolved in water in a calorimeter, the temperature fell from 25.00oC to 21.56oC. If the heat capacity of the solution and the calorimeter is 1071 J/oC,...
-
How is the standard form of a circles equation obtained from its general form?
-
In each case below, accounting conventions may have been violated. 1. After careful study, Lipski Company, which has offices in 40 states, has determined that its method of depreciating office...
-
For each of the following situations, use the weighted average method to determine the equivalent units of production for labor and overhead, assuming that they are continuously at the same...
-
Purina entered in a contract with the defendant to sell the defendant piglets, known as weanlingsbaby pigs that have been weaned. It is uncontested that the buyer breached and that Purina is entitled...
-
Carmin Kovach is single and has two children from her previous marriage. Anika, 9, lives with Carmin. Julius, 11, lives with his father, Ray. Carmin pays alimony of $400 per month to Ray. The...
-
During the early 19th century, the Springfield (Massachu-setts) Armory provided a good example of: an increasing division of labor accounting techniques for wage payments and control of time and...
-
Sulfur trioxide reacts with water to form sulfuric acid, a major contributor to acid rain. One origin of SO 3 is the combustion of sulfur, which is present in small quantities in coal, according to...
-
Write an SQL query that would ask the database to count the number of different types of t-shirts available and total their price. Your output should show only the field names: NumberOfTshirts...
-
Why is it advisable to set up a level with all three tripod legs on, or in, the same material (concrete, asphalt, soil), if possible?
-
""Q6: How long did the movie Gladiator aim to keep your attention? Save the series with this information in a variable called 'gladiator_duration', then print it. www
-
Do you agree or disagree? Explain why as w Proposition: RESOLVED: The State of California should legalize online sports betting Search entries or author Unread
-
Researchers have analyzed situation strength in all of the following elements EXCEPT O A. consequences OB. constraints OC. controllability O D. clarity O E. consistency ...
-
Alice drops a watermelon from the observation deck of a building 100 m above the ground. Standing directly below the watermelon, Bob fires an arrow straight up at 80 m/s at the same time Alice drops...
-
c. Suppose the CFO wants you to do a scenario analysis with different values for the cost savings, the machine's salvage value, and the net operating working capital (NOWC) requirement. She asks you...
-
Multi bank is a large London bank with a branch office in Boston. The American government believes that a prominent American underworld figure, Mr. Z, has been depositing stolen money in both the...
-
You continue to work in the corporate office for a nationwide convenience store franchise that operates nearly 10,000 stores. The per- store daily customer count (i.e., the mean number of customers...
-
Discuss how the DebyeHckel screening length changes as the (a) Temperature (b) Dielectric constant, (c) Ionic strength of an electrolyte solution are increased.
-
Why is it not possible to measure the Gibbs energy of solvation of Cl directly?
-
Why are activity coefficients calculated using the DebyeHckel limiting law always less than one?
-
Kleslia, Inc. budgeted manufacturing overhead of $365,224 at a level of 9,764 direct labor hours. The actual results at the end of February showed 2,338 direct labor hours and total manufacturing...
-
Explain the difference between Design effectiveness and operating effectiveness What is AS 2201? - It encourages the audit team to use the internal auditors in some situations, only after the audit...
-
Create an thread to discuss the following ideas: The different aspects of variable costs, fixed costs, and mixed costs.(Consider how they behave in regards to both total costs and unit costs.)...

Study smarter with the SolutionInn App


