Chromium can be detected in atomic absorption spectroscopy by monitoring the absorbance of UV light at a
Question:
Chromium can be detected in atomic absorption spectroscopy by monitoring the absorbance of UV light at a wavelength of 357.8 nm. What is the energy of a photon of this light?
Strategy We know the connection between photon energy and wavelength, which is given by Equation 6.3. Again, care with units requires conversion from nanometers to meters.
Equation 6.3.
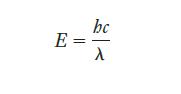
Transcribed Image Text:
E = bc λ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Analyze Your Answer The result is a very small number But we should realize that P...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
A helium-neon laser emits laser light at a wavelength of 632.8 nm and a power of 2.3 mW. At what rate are photons emitted by this device?
-
The laser used in cornea surgery to treat corneal disease is the excimer laser, which emits ultraviolet light at a wavelength of 193 nm in air. The index of refraction of the cornea is 1.376. What...
-
The active medium in a particular laser that generates laser light at a wavelength of 694 nm is 6.00 cm long and 1.00 cm in diameter. (a) Treat the medium as an optical resonance cavity analogous to...
-
Tin - Can, Inc. Aircraft ( TCAI ) R&D Project Management Problem Your group is hired to help TCAI Project Manager to solve the following problem. Using the activity time estimates and activity...
-
Thome Company uses a flexible budget for manufacturing overhead based on direct labor hours. Variable manufacturing overhead costs per direct labor hour are as follows. Indirect labor ........ $1.00...
-
Simons Company currently manufactures one of its crucial parts at a cost of $2.72 per unit. This cost is based on a normal production rate of 40,000 units per year. Variable costs are $1.20 per unit,...
-
Describe the six categories of early warning signs of potential fraud.
-
Donald Transport assembles prestige manufactured homes. Its job-costing system has two direct-cost categories (direct materials and direct manufacturing labor) and one indirect-cost pool...
-
(20 pts) Design a combinational circuit that counts ones at its inputs. The circuit has three 1-bit binary inputs (x, y, z) and two 1-bit binary outputs (f2, f1). "f2f1" is "00" if none of the inputs...
-
Unlike XRF, AAS cannot be used for nondestructive testing. Explain why not.
-
Consider a room that is 14 ft 20 ft with an 8-ft ceiling. (a) How many molecules of air are present in this room at 20C and 750 torr? (b) If a pollutant is present at 2.3 ppm, how many pollutant...
-
Let F(x) = x 0 sin t dt/t. Show that Evaluate F(1) to three decimal places. F(x) = x - x 3.3! + x5 5.5! x7 7.7! +
-
The effect of the new vouchers being offered by the state of Florida, will greatly impact the market of private schools. Considering the pre-voucher price was at equilibrium with both supply and...
-
An apartment building was purchased for $1,000,000 and was sold 5 years later at $1,200,000. During its holding period it produced 100,000 of profit each year. What is the HPR (before tax)?
-
1) Housing Prices in Melbourne discuss some of the welfare implications (think of consumer and producer surplus) of changes in housing prices.
-
3. Find a formula A containing three atoms p, q, and r, with the following property: for every assignment v: {p,q,r}{T, F} changing any of the values of v(p), v(q), v(r) will also change v(A).
-
Projects don't always go as planned; some unforeseen events may disturb the plan. Hence, it's important to identify risks early in the project life cycle, and risk identification should be a topic of...
-
The following table shows the last six years average new weekly unemployment insurance claims. Year Average weekly new unemployment insurance claims 2004 .............341,300 2005...
-
What exactly is a prima facie duty? How does an ethic of prima facie duties differ from monistic and absolutist ethical theories?
-
Compound A has molecular formula C 9 H 8 O 2 and exhibits a strong signal at 1740 cm -1 in its IR spectrum. Treatment with two equivalents of LAH followed by water gives the following diol. Identify...
-
Rank each set of compounds in order of increasing acidity: a. b.
-
Malonic acid has two acidic protons: The pKa of the first proton (pK 1 ) is measured to be 2.8, while the pK a of the second proton (pK 2 ) is measured to be 5.7. (a) Explain why the first proton is...
-
What is true for Inter-Vivos Trust and Testamentary Trust?
-
A baker's recipe for donuts yields 10 dozen donuts and it costs the baker $28.80 to produce the recipe. The raspberry filling the baker will use to fill the 10 dozen donuts costs the baker $7.13 to...
-
On November 15, 2023, Parkat Ltd., acquires control of Sparkat Ltd. Parkat and Sparkat are unrelated persons prior to the AOC. On November 14, 2023, Sparkat Ltd. owns the following properties: Land...

Study smarter with the SolutionInn App


