Consider the structure shown below for N 2 O 3 as well as any other important resonance
Question:
Consider the structure shown below for N2O3 as well as any other important resonance structures.
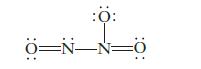
(a) What is the expected O—N—O bond angle in this structure?
(b) The N2O3 molecule contains N—O bonds of two different lengths. How many shorter N—O bonds would be present?
Transcribed Image Text:
:0: Ö=N- 0=N_N=0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a 120 b There will be one shorter bond The d...View the full answer

Answered By

Parvesh Kumar
I am an experienced Mathematics and Statistics tutor with 10 years of experience teaching students and working professionals. I love teaching students who are passionate to learn subjects or wants to understand any mathematics and statistics concept at graduation or master’s level. I have worked with thousands of students in my teaching career. I have helped students deal with difficult topics and subjects like Calculus, Algebra, Discrete Mathematics, Complex analysis, Graph theory, Hypothesis testing, Probability, Statistical Inference and more. After learning from me, students have found Mathematics and Statistics not dull but a fun subject. I can handle almost all curriculum of mathematics. I did B.Sc (mathematics), M.Sc (mathematics), M.Tech (IT) and am also Gate (CS) qualified. I have worked in various college and school and also provided online tutoring to American and Canadian students. I look forward to discussing with you and make learning a meaningful and purposeful
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Bridge to College (BTC) is a not-for-profit in the small college town of University Station which encourages undergraduates to form mentor relationships with local youth. The organization heavily...
-
Good Morning Food, Inc. is using the profitability index (PI) when evaluating projects. You have to find the PI for the company's project, assuming the company's cost of capital is 14.29 percent. The...
-
Now suppose this project has an investment timing option, since it can be delayed for a year. The cost will still be $70 million at the end of the year, and the cash flows for the scenarios will...
-
Walmart is one of the leaders in promoting the development and use of radio frequency identification (RFID) and electronic product codes. Explain how Walmart uses RFID in its procurement and...
-
What are some of the nonverbal clues to deception?
-
The comparative balance sheet of Portable Luggage Company at December 31, 2008 and 2007, is as follows: An examination of the income statement and the accounting records revealed the following...
-
Find the missing side lengths (in kilometers). (The sketches are not to scale.) 33 km 24 km 48 89 x 15 km 48% 89 22 km X = y = km E E km
-
Electrical engineers often use lithium niobate, LiNbO 3 , in designs for surface acoustic wave filters in devices such as cellular phones. What type(s) of bonding would you expect to be present in...
-
Consider the hydrocarbons whose structures are shown below. Which of these molecules would be planar, meaning that all of the atoms must lie in the same plane? Explain your answer in terms of orbital...
-
Let the random variable X follow a normal distribution with m = 50 and s2 = 64. a. Find the probability that X is greater than 60. b. Find the probability that X is greater than 35 and less than 62....
-
ABC Inc. borrows money at 9%, sells bonds at 6%, and the purchasers of common stock require 9% rate of return. If the company has borrowed $40 million, sold $60 million in bonds, and sold $100...
-
Often times, it is necessary to remove certain entries from a data set, list, etc., then create a new file or list with those results. a. Use a file created by your Lab 4 data file creator, which has...
-
Galaxy Corp. has to choose between two mutually exclusive projects. If it chooses project A, Galaxy Corp. will have the opportunity to make a similar Investment in three years. However, if it chooses...
-
A farmer has 34 less roosters than hens. If he has a total of 130 chickens altogether, how many roosters does he have?
-
An economy has two firms. Firm A produces wheat and firm B produces bread. In a given year, produces 50 bushels of wheat, sells 20 bushels to firm B at $3, exports 25 bushels at the same price to...
-
You are the new accounting manager at the Barry Transport Company. Your CFO has asked you to provide input on the company's income tax position based on the following: 1. Pretax accounting income was...
-
For the following exercises, write the polynomial function that models the given situation. Consider the same rectangle of the preceding problem. Squares of 2x by 2x units are cut out of each corner....
-
As the principal quantum number n increases, the electron is more likely to be found far from the nucleus. It can be shown that for H and for ions with only one electron such as He + , Calculate the...
-
In this problem, you will calculate the probability of finding an electron within a sphere of radius r for the H atom in its ground state. a. Show, using integration by parts, b. Using this result,...
-
The radius of an atom r atom can be defined as that value for which 90% of the electron charge is contained within a sphere of radius r atom . Use the formula in P 20.12b to calculate the radius of...
-
You are analyzing three stocks. Stock A has an expected return of 5%. Stock B has an expected return of 12%. Stock C has an expected return of 16%. You have the following variance-covariance matrix....
-
My objective identify the participants and the rules of an auction along with the items being auctioned. I learned that in a sealed-bid first-price auction, the high bidder wins and pays his value....
-
Explain each of the three characteristics of a market structure and how the market/industry you chose relates to each of these characteristics. Remember, all Market Structures are defined by these...

Study smarter with the SolutionInn App


