For the reaction of nitrogen, N 2 , and hydrogen, H 2 , to form ammonia, NH
Question:
For the reaction of nitrogen, N2, and hydrogen, H2, to form ammonia, NH3, a student is attempting to draw a particulate diagram, as shown below. Did the student draw a correct representation of the reaction? If not, what was the error the student made?
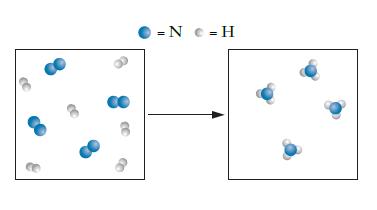
Transcribed Image Text:
= N c = H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
No the drawing is not correct ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
When your sympathetic nervous system is not activated and you don't have super- human powers, which hemisphere of your brain do you think you rely on most to process information? Under what...
-
Nitrogen (N2) and hydrogen (H2) react to form ammonia (NH3). Consider the mixture of N2 and H2 shown in the accompanying diagram. The blue spheres represent N, and the white ones represent H. Draw a...
-
For the reaction of nitrogen monoxide, NO, with chlorine, Cl2, 2NO(g) + Cl2(g) 2NOCl(g) the observed rate law is Rate = k[NO]2[Cl2] What is the reaction order with respect to nitrogen monoxide? with...
-
A money market portfolio has a market value of $20,000,000 and its value will change by $500 for a change in short-term yields of one basis point. The eurodollar futures contract has a tick size of...
-
The yields for Treasuries with differing maturities on a recent day were as shown in the table. a. Use the information to plot a yield curve for this date. b. If the expectations hypothesis is true,...
-
Describe the key factors contributing to effective cash management within a firm. Why is the cash management process more difficult in a MNC?
-
Plaintiff purchases a new car that has defects in its paint job. Three times the dealership repaints the care, but to no avail. The plaintiff continues to drive the car as he has no other option in...
-
Price and efficiency variances, journal entries. The Monroe Corporation manufactures lamps. It has set up the following standards per finished unit for direct materials and direct manufacturing...
-
Write a program in c to detect if the system will face any deadlock in the future. If a deadlock is detected then print "Deadlock Ahead" otherwise print "Safe here". The situation is given below....
-
The picture shown depicts the species present at the start of a combustion reaction between methane, CH 4 , and oxygen, O 2 . (a) Draw the resulting state after this set of reactants has reacted as...
-
Carbon dioxide is just one of many greenhouse gases in the atmosphere. What property makes a gas a greenhouse gas?
-
What was Kellys defense?
-
Sunland Candle Supply makes candles. The sales mix (as a percentage of total dollar sales) of its three product lines is birthday candles 30%, standard tapered candles 65%, and large scented candles...
-
9a. What is Bandwidth of an Analog signal that have 5 frequencies 100, 300, 500, 800, 900 Hz. (5) 9b. What is the phase of an analog signal that is offset by 1/8 cycle. (5 pt.) 10. A TCP segment with...
-
Explain why puffery is excluded from advertising law that states the only product claims that can be considered deceptive are those that are factually false.
-
Please use musical terms to describe the basic patterns this prelude is based on Bach Prelude and Fugue number two Tempered Clavier Book one with Harmonic Pedal.
-
Calculate the weighted average cost of capital for the following firm assuming Fama-French 3 factor model (market factor risk premium = 7%; size factor risk premium = 3.7%; and book-to-market factor...
-
Your manager requires that you, as cashier, immediately enter each sale. Recently, lunch hour traffic has increased and the assistant manager asks you to avoid delays by taking customers' cash and...
-
In the current year, the City of Omaha donates land worth $500,000 to Ace Corporation to induce it to locate in Omaha and create an estimated 2,000 jobs for its citizens. a. How much income, if any,...
-
Propose a plausible mechanism for the following process, called iodolactonization: I2
-
When 3-bromocyclopentene is treated with HBr, the observed product is a racemic mixture of trans-1,2-dibromocyclopentane. None of the corresponding cis-dibromide is observed. Propose a mechanism that...
-
Provide a systematic name for each of the following compounds: (a) (b) (c) (d)
-
Q1: Sponge and Bob-Group consolidated SFP Below are the statements of financial position of two companies as at 31 December 2020 Sponge Pic 000 Bob Pic C000 Non-cument assets Property, plant and...
-
Consider the bonds below, all of which pay coupons semi-annually and so in each case the yield to maturity is a semi-annual APR. As- sume that each bond has face value equal to $1,000 and that in...
-
Suppose CAPM assumptions hold. Suppose you are given the following regarding two assets A and B traded in a market M: Er=10.5% Erg=13% and and BA=0.7 BB=1.2 Answer the following questions: 1. [2]...

Study smarter with the SolutionInn App


