Look at the table of electron configurations in Appendix c. Which elements have configurations that are exceptions
Question:
Look at the table of electron configurations in Appendix c. Which elements have configurations that are exceptions to the aufbau principle? Propose a reason why these elements have these exceptions.
Data from appendix c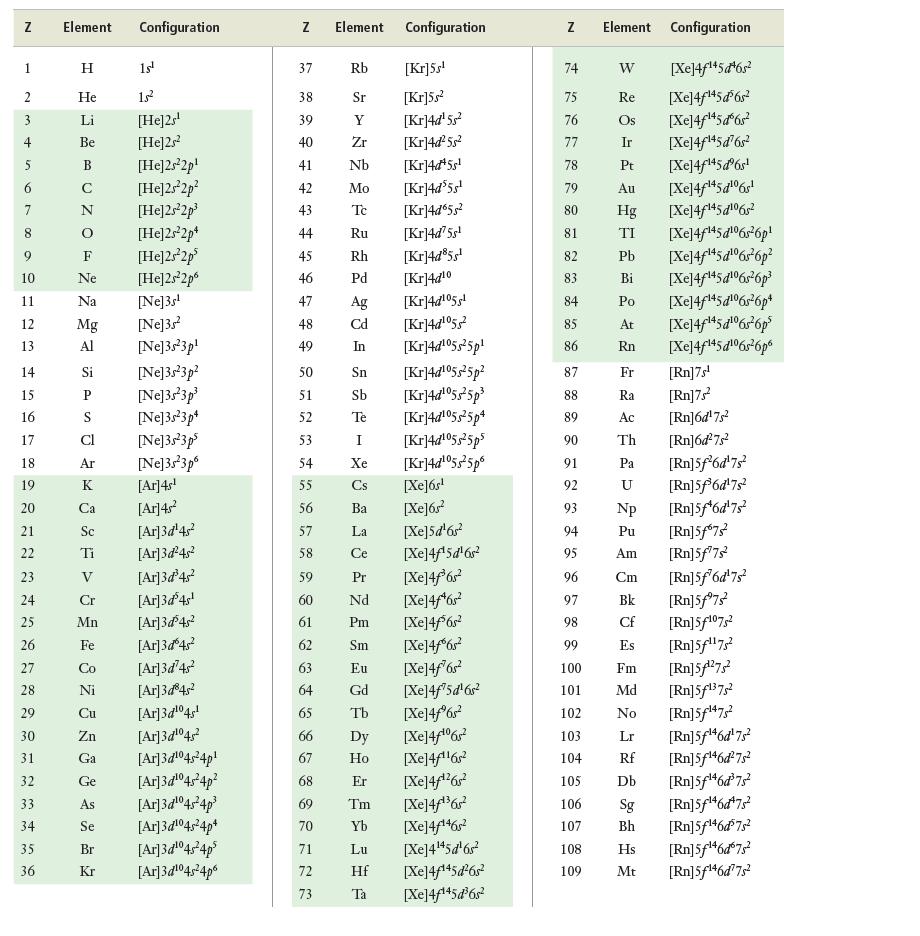
Transcribed Image Text:
N 1 2 + 5 6 7 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 Element Configuration H 15¹ He 15² Li [He]2s¹ Be [He]2s² [He]2s²2p¹ [He]2s²2p² [He]2s²2p³ [He]2s²2p* [He]2s²2p³ [He]2s²2p6 [Ne]3s¹ [Ne]3s² [Ne]3s²3p¹ AUZO Z Z Ne Na Mg Al Si P S Ar K Ca Sc Ti V Cr Mn Fe Co [Ne]3s²3p² [Ne]3s 3p³ [Ne]3s²3p4 [Ne]3s 3p5 [Ne]3s²3p6 [Ar]4s¹ [Ar]4s² [Ar]3d¹4s² [Ar]3d²4s² [Ar]3d³4s² [Ar]3d³4s¹ [Ar]3d³45² [Ar]3d 4s² [Ar]3d¹4² Ni [Ar]3d³45² Cu [Ar]3d¹⁰45¹ Zn [Ar]3d¹⁰45² Ga [Ar]3d¹04s²4p¹ Ge [Ar]3d¹⁰45²4p² As [Ar]3d¹04s²4p³ Se [Ar]3d¹04s²4p+ Br [Ar]3d¹04s²4p³ Kr [Ar]3d¹04s²4p6 N 37 38 39 40 41 42 43 44 45 46 7 48 49 50 47 50 Element Configuration 72 75 Rb [Kr]5s¹ Sr [Kr]55² Y [Kr]4d¹5s² Zr [Kr]4d²5s² Nb [Kr]4d¹5s¹ Mo [Kr]4d³5s¹ Tc [Kr]4d65s² Ru [Kr]4d' 5s¹ Rh [Kr]4d³5s¹ Pd [Kr]4d10 [Kr]4d¹05¹ [Kr]4d¹055² [Kr]4d¹055²5p¹ [Kr]4d¹05s²5p² [Kr]4d¹05s²5p³ [Kr]4d¹05525p4 [Kr]4d¹05s²5ps [Kr]4d¹055²5p6 73 Ag Cd In 51 52 53 54 55 56 Ba 57 La 58 Ce 59 Pr 60 Nd 61 Pm 62 Sm 63 Eu 64 65 Sn Sb Te [Xe]6s¹ [Xe]6s² [Xe]5d¹6s² [Xe]4f¹5d¹6s² [Xe]4far [Xe]4f¹6s² [Xe]4f6r2 [Xe]4f6s? [Xe]4f762 [Xe]4f¹5d¹6s² [Xe]4f®6? 66 Dy [Xe]4/06r 67 Ho [Xe]4f1162 68 Er [Xe]4f*26r? 69 Tm [Xe]4f36r2 70 Yb [Xe]4f1462 71 Lu [Xe]4145d6r Hf [Xe]4f¹45d²6s² Ta [Xe]4f¹45d³6s² I Xe Cs Gd Tb Z 74 75 76 77 78 79 80 81 82 83 84 85 86 87 88 89 90 91 92 93 94 95 96 97 98 99 100 101 102 103 104 105 106 107 108 109 Element W Re Os Ir Pt Au Hg TI Pb Bi Po At Rn Rf Db Configuration [Xe]4f145 đ6g? [Xe]4f14562 [Xe]4/145d6s? [Xe]4f145d6s? [Xe]4f¹45dº6s¹ [Xe]4f**5¢06r! [Xe]4f145g16g? Sg Bh [Xe]4f45d!06r6p! [Xe]4f**5d!06r6p2 [Xe]4f145¢106s6p* [Xe]4f145¢106s26p* [Xe]4f**5¢!6r6p* [Xe]4f**5d106r6p Fr Ra Ac Th Pa U Np Pu Am Cm Bk Cf Es Fm Md [Rn]5f¹³75² No [Rn]5f¹47² Lr [Rn]5f¹46d¹75² [Rn]5f¹46d²75² [Rn]5f¹46d³75² [Rn]5f¹46d75² [Rn]5f¹46d³75² Hs [Rn]5f¹46d675² Mt [Rn]5f¹46d775² [Rn]7s¹ [Rn]7s² [Rn]6d¹7s² [Rn]6d²7s² [Rn]5f6d¹7s [Rn]5f³6d¹7s² [Rn]5f¹6d¹7s² [Rn]5f673² [Rn]5f¹7s² [Rn]5f6d¹7s² [Rn]5f7s² [Rn]5f¹07,² [Rn]5f¹¹75² [Rn] 5¹27²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The elements that have configurations that are exceptions to the ...View the full answer

Answered By

Ariz Azad
I love explaining things to people. Sharing my knowledge with others is one of the rare things that gives me immense pleasure. I have been a tutor on Chegg for more than a year now for Finance. Nothing beats the satisfaction of getting a thumbs up from students for your answer. Maintaining quality in teaching is one of the things I have learned through experience.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
The CICA Handbook is available to most post-secondary students through their institutions subscription. Visit edu.knotia.ca to complete this exercise. (You may need to complete this exercise on...
-
Look again at the table of post-graduation plans for the senior class in Exercise 27. a) Find the conditional distributions (percentages) of plans for the white students. b) Find the conditional...
-
Write a C++ program with recursion to print the following pattern for any odd number greater than or equal to 5. Note: the last row must be filled with digits from your ID in case the pattern is...
-
Presto Corp. has collected the following data concerning its maintenance costs for the past 6 months. Compute the variable and fixed cost elements using the high-lowmethod. July August September...
-
The following comment appeared in the notes of Alvarado Corporations annual report: Such distributions, representing proceeds from the sale of James Buchanan, Inc. were paid in the form of partial...
-
An engineer prepares a report to evaluate a project using PW and IRR. Just before submitting the report, he spills coffee on it, making the first digit of the 2-digit IRR unreadable. The second digit...
-
The following data are taken from the general ledger and other records of Coral Park Production Co. on January 31, the end of the first month of operations in the current fiscal year:...
-
What is the Federal Budget? Explain the process used by the government to create the budget. How does this process differ from the process described by your interviewee who you interviewed in week...
-
Distinguish between the terms core electrons and valence electrons.
-
From the list of atoms and ions given, identify any pairs that have the same electron configurations and write that configuration: Na + , S 2-, Ne, Ca 2+ , Fe 2+ , Kr, I - .
-
Blackwell Automotive's balance sheet at the end of its most recent fiscal year shows the following information: In addition, it was reported that the firm had a net income of $156,042 on sales of...
-
How would you like to differentiate standard unqualified audit report and standard qualified audit report.
-
what other services you can use to get more information and updates on tax issues affecting your clients. What can small businesses do to keep up with ever-changing tax laws? Where do you draw the...
-
Using God's law or intent: can you give me some examples having to do with property or wealth?
-
What is a trust structure? Does the trust structure pay tax? Does this seem unusual?
-
Why was the Barangay Micro business Enterprises (BMBE) of the Republic act 9178 created? What is their reason?
-
Consider a project with net benefits NBt in period t: NB0 = 30, NB1 = 15, NB2 = 25, NB3 = 3. a. Find the net present value if the discount rate is r = 6%, 8% and 10%. b. Now assume that the discount...
-
A line l passes through the points with coordinates (0, 5) and (6, 7). a. Find the gradient of the line. b. Find an equation of the line in the form ax + by + c = 0.
-
Identify which of the following compounds is expected to have the larger heat of combustion:
-
Draw each of the following compounds: (a) 2,2,4-Trimethylpentane (b) 1,2,3,4-Tetramethylcycloheptane (c) 2,2,4,4-Tetraethylbicyclo [1.1.0] butane
-
Sketch an energy diagram that shows a conformational analysis of 2,2-dimethylpropane. Does the shape of this energy diagram more closely resemble the shape of the energy diagram for ethane or for...
-
What is your current age? How much do you need to put in an account starting today to have $1 million at age 62? Answer text Question 7 Rich text editor
-
5. You are considering the following two mutually exclusive projects. The crossover rate between these two projects is percent and Project should be accepted if the required return is greater than...
-
If a universal life insurance policy uses "adjustable monthly mortality deduction rates," what does that usually mean? Question 4 options: The rate charged for the insurance element of the contract...

Study smarter with the SolutionInn App


