The molecule shown below is responsible for the smell of popcorn. Write the correct molecular formula for
Question:
The molecule shown below is responsible for the smell of popcorn. Write the correct molecular formula for this compound.
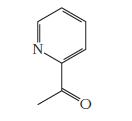
Transcribed Image Text:
N.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The molecule depicted appears to be that of a subst...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
The molecular formula of allicin, the compound responsible for the characteristic smell of garlic, is C6H10OS2. (a) What is the molar mass of allicin? (b) How many moles of allicin are present in...
-
The molecule shown below is called furan. It is represented in typical shorthand way for organic molecules, with hydrogen atoms not shown. (a) What is the molecular formula for furan? (b) How many...
-
Show that Hint: Recall that and tPx = e lo Uxtu du fx (t) = == -Sx (t), dt fx(t) = tPx Hx+t.
-
Cost information for Lake County Library is as follows. In addition to directly traceable costs, the library incurred $24,000 for a building lease. REQUIRED A. Allocate to departments any costs that...
-
The Gansac Publishing Company signed a contract with an author to publish her book. The signing took place on January 1, 2007 and a payment of $20,000 was made. The agreement was that the author...
-
Gunderson Lutheran Health System in La Crosse, Wisconsin, is a health care delivery company that includes a 325-bed hospital, several specialty medical practices, and 41 clinics. In a recent year,...
-
Ormet Primary Aluminum Corporation, operated an aluminum smelter plant in Hannibal, Ohio. The facility ceased production in October 2013 in order to liquidate its assets after filing for bankruptcy...
-
Valdespin Company manufactures three sizes of camping tentssmall (S), medium (M), and large (L). The income statement has consistently indicated a net loss for the M size, and management is...
-
Provide an objective setting paragraph on HP Inc.'s decision to increase the R&D investment to use towards their partnership with Poly Solutions to enhance Microsoft Teams flexibility
-
What is a functional group? How does the concept of the functional group help to make the study of organic chemistry more systematic?
-
What is meant by the phrase organic chemistry?
-
A company is testing a new medicine for migraine headaches. In the study, 150 women were given the new medicine and an additional 100 women were given a placebo. Each participant was directed to take...
-
A simple camera telephoto lens consists of two lenses. The objective lens has a focal length f = +42.0 cm. Precisely 34.8 cm behind this lens is a concave lens with a focal length f = -13.8 cm. The...
-
Problem # 7 Describe the ElGamal public key cipher and design an example of the cipher "for small numbers" with an example of ciphering. Problem # 8 Give an example proving that the assumption...
-
Which shell command can you use to display information about mounted file systems? 1 1 point cat ps df head
-
12 lbm of a gas occupies a volume of 48 ft at sea level. Calculate the following properties for the gas and include the appropriate units. a) Specific volume (v) b) Density (p) c) Specific weight (y)
-
Two identical rocks are thrown horizontally from a cliff with different velocities. The rocks are thrown at the same time and are shown below while they are still in the air after a few seconds. For...
-
Northwest Minerals operates a mine. During July, the company obtained 500 tons of ore, which yielded 250 pounds of gold and 62,500 pounds of copper. The joint cost related to the operation was...
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
Each of the following molecules has one plane of symmetry. Find the plane of symmetry in each case: a. b. c. d. e. f. CI Me Me Br
-
Compound A has molecular formula C5H12, and monobromination of compound A produces only compound B. When compound B is treated with a strong base, a mixture is obtained containing compound C and...
-
Compare the Lewis dot structure of nitrogen and phosphorus and explain why you might expect these two atoms to exhibit similar bonding properties?
-
How do our biases affect decision making? Think about the topic of ethics in light of the decision-making biases mentioned. Can our biases "cause" unethical behavior? Why or why not?
-
How do you currently or how will you be your student's champion? The introduction of The Listening Leader lists what a transformational school looks like (xxx). Do you agree or disagree with this...
-
Your house is up for a rent review. You know that the rental rates in your area have gone up by about 20%. You expect your landlord to demand for an escalation in rent. You are keen to continue in...

Study smarter with the SolutionInn App


