Use the standard reduction potentials for the reactions: to calculate the value of Ksp for silver chloride
Question:
Use the standard reduction potentials for the reactions:
![]()
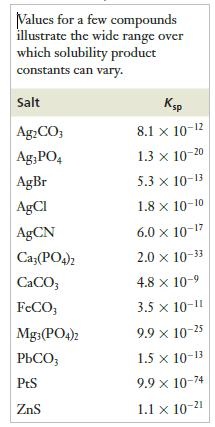
to calculate the value of Ksp for silver chloride at 298 K. How does your answer compare with the value listed in Table 12.4?
Table 12.4
Transcribed Image Text:
AgCl(s) +eAg(s) + Cl (aq) and Ag+ (aq) + e→Ag(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
K sp 17 10 10 T...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Use the standard reduction potentials to find the equilibrium constant for each of the following reactions at 25°C: (a) (b) (c) Br2(1) + 21-(aq )- 2Br_(aq) + 12(s) 5Fe2 + (aq) +MnO4 (aq ) + 8H +...
-
The standard reduction potentials of the following half-reactions are given in Appendix E: (a) Determine which combination of these half-cell reactions leads to the cell reaction with the largest...
-
Using the standard reduction potentials listed in Appendix E, calculate the equilibrium constant for each of the following reactions at 298 K: (a) Cu(s) + 2 Ag+ (aq) Cu2+ (aq) + 2 Ag(s) (b) 3 Ce4+...
-
MOSS COMPANY Income Statement \ table [ [ , ] , [ Sales For Year Ended December 3 1 , 2 0 2 1 , $ 5 2 5 , 0 0 0 es MOSS COMPANY Income Statement For Year Ended December 31, 2021 Sales Cost of goods...
-
List five or more topics that an organization might feature in a press release.
-
The following selected information was extracted from the 20x1 accounting records of Lone Oak Products: Raw material purchases ................................ $ 175,000 Direct labor...
-
Using the data in question 4, Department Xs contribution to overhead as a percentage of sales is a. 20%. c. 12%. e. 32%. b. 30%. d. 48%. Data From Question 4 A company operates three retail...
-
The Walton Toy Company manufactures a line of dolls and a doll dress sewing kit. Demand for the dolls is increasing, and management requests assistance from you in determining an economical sales and...
-
Q1) Find expressions for the Revenue, Cost, and Profit from selling x thousand items. Item Price Variable Cost $8.00 3816x Fixed Cost $71,174
-
Calculate the equilibrium constant for the following reactions using data from the standard reduction potential tables. (a) Cl(g) + 2 Br (aq) Br(g) + 2 Cl(aq) (b) Ni(s) + 2 Ag+ (aq) 2 Ag(s) + Ni+...
-
The equilibrium constant for a reaction is 3 10 15 . (a) Without carrying out any calculation, discuss whether G for the reaction is positive or negative. (b) Calculate G for this reaction.
-
Do you think career development can adversely affect organizational performance in that the process sometimes convinces the involved parties to change jobs?
-
The population of a herd of deer is represented by the function A (t) = 190(1.21)t, where t is given in years. To the nearest whole number, what will the herd population be after 4 years? The herd...
-
Current Attempt in Progress Vermillion Ltd. prepares an aging schedule for its accounts receivable at the end of each month and records credit losses monthly. The following selected information is...
-
Acme Medical Supply Company desires a target operating income amount of $100,000, with assumption inputs as follows: Desired (target) operating income amount = $100,000 Unit price for sales = $80...
-
Consider accounting firm guidance-what additional interpretation / examples can you find that address this issue? Using the Conceptual Framework, evaluate whether this percentage rental change meets...
-
Mahalo Boat Adventure Inc. has a July 31 year-end. It showed the following partial amortization schedules regarding two bond issues: Bond Issue A $ 26,600 Period Ending June 1/20 Dec. 1/20 (A) Cash...
-
Generally, final design results are rounded to or fixed to three digits because the given data cannot justify a greater display. In addition, prefixes should be selected so as to limit number strings...
-
Access the Federation of Tax Administrators Internet site at www. taxadmin.org/state-tax-forms and indicate the titles of the following state tax forms and publications: a. Minnesota Form M-100 b....
-
The truss is made of three A-36 steel members, each having a cross-sectional area of 400 mm 2 . Determine the horizontal displacement of the roller at C when P = 8 kN. 5 kN 0.8m 0.8 m. 0,6 m-
-
The A-36 steel drill shaft of an oil well extends 12 000 ft into the ground. Assuming that the pipe used to drill the well is suspended freely from the derrick at A, determine the maximum average...
-
The 2014-T6 aluminium rod has a diameter of 30 mm and supports the load shown. Determine the displacement of end A with respect to end E. Neglect the size of the couplings. 8 kN 2 kN 6 kN 4 kNY -2m-...
-
Consider the following cash budgeting example for Buckeye Pharmaceutical Company: Gross Sales by month: . May 2010: $5,000 . June: $5,000 . July: $10,000 . August: $15,000 . September: $20,000 ....
-
Question #10 Risk/Variance & Mergers Part 1: Answer each of the five (5) True/False questions below (1 point each): A. Portfolio variance will never be greater than the investment with greatest...
-
PLEASE ANSWER ACCURATELY Safari File Edit View History Bookmarks Window Help cord ... Dashboard Logout M Inbox (6,406... Simple Inter... < > ABP bbhosted.cuny.edu Fall 2013 - S... Compound I......

Study smarter with the SolutionInn App


