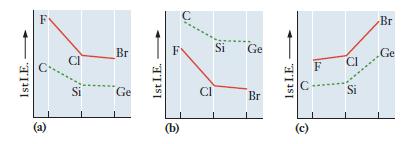
Which graph correctly depicts the first ionization energy of three elements in groups 14 (dashed line) and
Question:
Which graph correctly depicts the first ionization energy of three elements in groups 14 (dashed line) and 17 (solid line)? Explain the reasoning you used to make your choice.
Transcribed Image Text:
1st I.E. Si Br Ge 1st I.E. (b) Si Ge Br 1st I.E. (c) F Br Ge
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
Graph b correctly depicts the first ionization energy of three elements in groups 14 dashed line and ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Graph f(x) - x, g(x) x +3 and (x) x5. Calculate the derivatives of f, g and h.
-
Explain why the first ionization energy tends to increase as one proceeds from left to right across a period. Why is the first ionization energy of aluminum lower than that of magnesium and the first...
-
The first ionization energy of the oxygen molecule is the energy required for the following process: O2 (g) O2+ (g) + e- The energy needed for this process is 1175kJ/mol, very similar to the first...
-
Ultimate Consulting is a management consulting firm that offers a wide range of services to companies in many industries. To stay on the cutting edge of developments in the business world and to...
-
Stephen Thorne owns the Fredonia Barber Shop. He employs five barbers and pays each a base rate of $1,000 per month. One of the barbers serves as the manager and receives an extra $500 per month. In...
-
Presented below are two independent situations. Situation 1 Conchita Cosmetics acquired 10% of the 200,000 shares of common stock of Martinez Fashion at a total cost of $13 per share on March 18,...
-
Many researchers are interested in the transcription of protein-encoding genes in eukaryotes. Such researchers want to study mRNA. One method that is used to isolate mRNA is column chromatography....
-
Tower Interiors has made the forecast of sales shown in the following table. Also given is the probability of each level of sales. Sales Probability $200,000 ........ 0.20 300,000 ......... 0.60...
-
7. In each of the following scenarios, a through d, determine whether the following is a microeconomic or macroeconomic issue and WHY. a. Dell computers announces that it will raise the price of its...
-
The visible lines in the hydrogen atom emission spectrum arise from transitions with a final state with n = 2. In what spectral region should we expect to find transitions that have a final state of...
-
A particular element has the following values for its first four ionization energies: 900, 1760, 14,850, and 21,000 kJ/mol. Without consulting a list of ionization energy values, determine what group...
-
The Johnson Company, a corporation organized under the laws of State X, after proper authorization by the shareholders, sold its entire assets to the Samson Company, also a State X corporation....
-
A taxpayer wishes to buy some electric vehicles for his business and get the Commerical Clean Vehicle Credit for his purchases when he files for TY2023. He plans on buying four (4) vehicles, only one...
-
Consumption under borrowing constraint (a) With borrowing constraint, household can borrow until 200 in period 1. Under y1 =100, y2= 200, r= 0.2, How much is maximum possible consumption of period 1?...
-
In each of Problems 1 through 4, determine "(xo), " (xo), and (4)(xo) for the given point to if y = o(x) is a solution of the given initial value problem. 1. y + xy + y = 0; y(0) = 1, 2. y + (sin...
-
A taxpayer operating a sole proprietorship and filing on a Schedule C pays $65,000 in health insurance premiums for himself and his employees. $15,000 of that amount is for premiums paid to cover...
-
4. Given the following for a carbide insert single point tool, what is the expected tool life for a high-speed operation given: . = 257.35 b. Vc = 10 m/sec c. So = 1mm/rev d. T = 3mm e. y = 0.2...
-
Smith Corp. has determined that its contribution margin , (P MC)/P, is 40%. A recent market research study found the following relationship between adverting outlays and sales revenue. Advertising...
-
Let (x) = x 2 - 9, g(x) = 2x, and h(x) = x - 3. Find each of the following. (((--) 2
-
Draw structures for all constitutional isomers with the molecular formula C 4 H 8 that have: a. Only single bonds b. One double bond
-
What is the relationship between a state function and an exact differential?
-
Is the following statement always, never, or sometimes valid? Explain your reasoning: H is only defined for a constant pressure process.
-
Critical Thinking 10-7: Dynamic Mobile Access There are so many opportunities now for people to allow constant and dynamic connection between their mobile phone and an external (often unregulated)...
-
In Matlab, create a code that puts the value three-halves pi radians, 3 ????, in a variable named "angle". Then set another variable named angleDegrees equal to a calculation that converts the value...
-
Following our lectures, what are the main components/approaches/procedures to cluster data? Describe where we use them and what are the advantages and disadvantages of them. Refer only to topics...

Study smarter with the SolutionInn App


