According to both the Bohr model and the quantummechanical model, the energy of the hydrogen atom can
Question:
According to both the Bohr model and the quantummechanical model, the energy of the hydrogen atom can be calculated from the quantum number n with the following equation: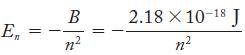
Express the energy, in joules, of the three lowest energy states of the hydrogen atom.
Transcribed Image Text:
En B n² 2.18 X 10-18 J n²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The given formula is E n21810 18 J n 2 Substitute n1 in the formula to find the energy of first ener...View the full answer

Answered By

Nandkumar Ghadge
From my childhood I was fascinated by this world. Knowledge is scattered around that you have to garnish and distribute among others and is to be used for welfare of mankind.
I have 4 years of teaching experience to undergraduate students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
LIST-I contains compounds and LIST-II contains reactions. LIST-I (1) HO (II) (III) (IV) Mg(OH)2 BaCl CaCO3 (P) (Q) (R) LIST-II Mg(HCO3) + Ca(OH) BaO,+H,SO4 Ca(OH) + MgCl BaO, +HCI (T) Ca(HCO3)2 +...
-
The Bohr model of the hydrogen atom states that the single electron can exist only in certain allowed orbits around the proton. The radius of each Bohr orbit is r = n2(0.052 9 nm) where n = 1, 2, 3,...
-
Which one of the following statements is false? (a) The orbits in the Bohr model have precise sizes, whereas in the quantum mechanical picture of the hydrogen atom they do not. (b) In the absence of...
-
Data-2-Go manufactures and sells flash drives. The company produces only when it receives orders and, therefore, has no inventories. The following information is available for the current month:...
-
A uniform rod of mass M = 20 kg and length L = 5 m is bent into a semicircle. What is the gravitational force exerted by the rod on a point mass m = 0.1 kg located at the center of the circular arc?
-
Peachtree Apartments, Inc.s unadjusted and adjusted trial balances at April 30, 2010, follow. Requirements 1. Make the adjusting entries that account for the differences between the two trial...
-
Rework Problem 10.14, part a, but exclude the region information. a. Comment on the difference in the models you have found. Is there indication that the region information substantially improves the...
-
The following income statements were drawn from the annual reports of Pierro Sales Company. The president's message in the company's annual report stated that the company had implemented a strategy...
-
Monie Love Inc. began operations in January 2 0 2 0 and had the following reported net income or loss for each of its 5 years of operations: 2 0 2 0 $ 2 2 5 , 0 0 0 loss 2 0 2 1 $ 1 4 0 , 0 0 0 loss...
-
Critically discuss whether the CAPM makes portfolio theory redundant.
-
Find the uncertainty in the position (in m) of a 650-kg automobile that is moving at 55 mph if the speed is known to within 1 mile/hr. Is this uncertainty in position significant?
-
The energy expression given for the allowed states in the hydrogen atom, -2.18 10 -18 J/n 2 , refers to a single atom. Express the energy of the allowed states (in kJ/mol).
-
What is the expected return and beta of your portfolio using the following data: Market risk premium = 8 percent Risk-free rate = 4 percent Beta of XYZ = 1.5; Beta of PDQ = 2.0 Investment in XYZ...
-
If your tires are worn, does your odometer (which is keyed to the turns of the axle) underestimate or overestimate the number of miles you drive?
-
The Foodpanda group is a worldwide mobile food delivery marketplace headquartered in Berlin, Germany. It was launched in March 2012. It allows you to select from local restaurants and place orders...
-
What is the formula for the rotational inertia of a thin spherical shell of inertia \(m\) and radius \(R\) about an axis tangent to any point on its surface?
-
For a given level of technology, there is a maximum density of "bits" of information that can be stored on a computer's hard-drive disk. The data are stored in a tight spiral, so that "tracks" are...
-
A gymnast's backflip is considered more difficult to do in the layout (straight body) position than in the tucked position. Why?
-
Use the values in the contingency table to solve the equations given. a. P(F A) = _________ b. P(A | B) = _________ c. P(B) = _________ d. P(E F) = _________ e. P(D | B) = _________ f. P(B | D) =...
-
Solve for the equilibria of the following discrete-time dynamical systems Pr pt+1 = Pr+2.0(I-Pr)
-
Calculate the rotational partition function for the interhalogen compound F 35 Cl (B = 0.516 cm 1 ) at 298 K.
-
Calculate the rotational partition function for 35 Cl 2 (B 0.244 cm -1 ) at 298 K.
-
For which of the following diatomic molecules is the high-temperature expression for the rotational partition function valid at 40. K? a. DBr (B = 4.24 cm 1 ) b. DI (B = 3.25 cm 1 ) c. CsI (B =...
-
How to complete these steps using this excel format from the image below. Yearly Sales January February March April May June July August September October November December 2015 2016 2017 2018 2019...
-
What would be the total fire insurance premium for Best Seconds if their $857,500 building belongs in structural classification D?
-
What did the Central Banks decided concerning the target interest rates or any other target they have? What was the justification for the decision? *Bank of Japan *Bank of England *Bank of Canada...

Study smarter with the SolutionInn App


