Assign the oxidation numbers of all atoms in the following species.
Question:
Assign the oxidation numbers of all atoms in the following species.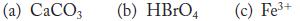
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
When assigning oxidation numbers to the atoms in a compound we follow a set of rules that help us de...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Assign the oxidation numbers of all atoms in the following species.
-
Assign the oxidation numbers of all atoms in the following compounds.
-
Assign the oxidation numbers of all atoms in the following compounds. (a) KHF 2 (b)H 2 Se (c)NaO 2 (d)C 2 H 6
-
Quality Air Conditioning manufactures three home air conditioners: an economy model, a standard model, and a deluxe model. The profits per unit are $63, $95, and $135, respectively. The production...
-
One scheme for extending the operation of gas turbine blades to higher temperatures involves applying a ceramic coating to the surfaces of blades fabricated from a super alloy such as inconel. To...
-
For someone with a foot length of 29.0 cm, identify the 95% prediction interval estimate of their height, and write a statement interpreting that interval. Refer to the Minitab display obtained by...
-
How do we use the requirement of design to define messages and methods?
-
Greener Grass Fertilizer Company plans to sell 200,000 units of finished product in July and anticipates a growth rate in sales of 5 percent per month. The desired monthly ending inventory in units...
-
What would be the gross debt service (GDS) ratio based on the following information? Current monthly gross income = $4000 Current monthly after-tax income = $2800 Monthly mortgage payment = $950...
-
Balance the following reactions, and specify which species is oxidized and which is reduced. (a) H 2 + O 2 H 2 O (b) Fe + O 2 Fe 2 O 3 (c) Al 2 O 3 + C Al + CO 2
-
Assign the oxidation numbers of all atoms in the following species.
-
What do you observe immediately and after 5 minutes? Prepare a table like the one below.
-
19. Explain modern securitized (shadow) banking . What roles do (1) Haircuts. (2) collateral, (3) repo rates and (4) loans securitized play and how do they compare to the role of banknote, collateral...
-
What are common law contracts and can common law contracts be verbal?
-
What are the origins of the common law? Name one or more Common Law principles that exist in our law today and explain them ?
-
Describe the following in 1. Describe the common law system. 2. Describe the differences between the common law system and the civil law system 3. Describe the meaning of precedent in the common law...
-
The most distinguishing feature of common law is the reliance on the doctrine of precedent when making court judgments. It is not restricted to judicial decisions generated within Hong Kong but case...
-
Why is it so important to specify the audit objective when planning a sample to test an account balance?
-
Use the graphs of f and g to graph h(x) = (f + g) (x). To print an enlarged copy of the graph, go to MathGraphs.com. 1. 2. y 24 8. 2. -2 -2 4 6
-
Two mischievous children drop water balloons from a bridge as depicted in Figure P3.87. Each water balloon is approximately 30 cm in diameter, and in this figure the red balloon is about 1.8 m below...
-
An impish young lad stands on a bridge 10 m above a lake and drops a water balloon on a boat of unsuspecting tourists. Although the boat is traveling at a speed of 7.5 m/s, the boy manages to land...
-
A boy pushes a 3.1-kg book against a vertical wall with a horizontal force of 40 N. What is the minimum coefficient of friction that will keep the book in place without sliding?
-
a. Excel Courier (Gh.) Ltd. has an 8% bond making semi-annual payments. The bond has a face value of GH1,000 and has 13 years to maturity. Aseda Co. Ltd. has just issued a 10-year 10% coupon bond...
-
5. (1.5 points) Suppose the yield to maturity on a one-year zero-coupon bond is 2%. The yield to maturity on a two-year zero-coupon bond is 4%. (a) According to the Expectations Hypothesis, what is...
-
Convert the following Annual Salaries (10 marks) Annually Monthly $55,000.00 $1058 $40,000.00 $7692 $37,500.00 $7211 $24,000.00 $4615 $96,000.00 $ Semi-Monthly Bi-Weekly Weekly I

Study smarter with the SolutionInn App


