At 3000 K, carbon dioxide dissociates 1 CO(g) CO(g) + -O(g) K, for this reaction is 2.48.
Question:
At 3000 K, carbon dioxide dissociates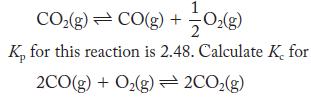
Transcribed Image Text:
1 CO(g) CO(g) + -O(g) K, for this reaction is 2.48. Calculate K, for 2CO(g) + O(g)2CO(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Given the equilibrium reaction COg COg O2g The equili...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
One mole of water and one mole of CO2 are placed in a reactor at 3000 K and a total pressure of 400 kPa. The water dissociates into hydrogen and oxygen, and the carbon dioxide dissociates into carbon...
-
Consider the equilibrium dissociation of carbon dioxide CO 2 CO + 1/2 O 2 . At 2500 K, the equilibrium constant is 0.03635. Calculate the enthalpy of reaction for this reaction at 2500 K and use...
-
Carbon dioxide gas enters a pipe at 3 MPa and 500 K at a rate of 2 kg/s. CO2 is cooled at constant pressure as it flows in the pipe and the temperature CO2 drops to 450 K at the exit. Determine the...
-
Neo-Darwinism believes that new species develop through (A).Continuous variations and natural selection (B) Mutation with natural selection (C) Hybridization (D) Mutation
-
Name the five steps in process costing when equivalent units are computed?
-
Google has changed its strategy with regard to how much and over which media it invests in advertising. The following table shows Googles marketing budget in millions of dollars for 2008 and 2011...
-
It is said that "the higher the MARR, the higher the price that a company should be willing to pay for equipment that reduces annual operating expenses." Explain the reasoning behind this statement.
-
In the market share analysis of Section 17.1, suppose that we are considering the Markov process associated with the shopping trips of one customer, but we do not know where the customer shopped...
-
InstaChat Company had a beginning retained earnings balance of $80,000 and an ending retained earnings balance of $60,000. In the current period the company had revenues of $110,000 and expenses of...
-
Exactly 10.0 mL of a 0.0502 M KCl solution is added to 20.0 mL of 0.0259 M AgNO 3 . (a) Write the net ionic equation for the reaction. (b)Calculate the mass of the insoluble product formed. (c)...
-
Find the concentration of silver necessary to begin precipitation of AgCl from a solution in which the Cl - concentration is 7.4 10 -4 M.
-
Unless stated otherwise, B is a basis for a vector space V. Justify each answer. (T/F) In some cases, a plane in R 3 can be isomorphic to R 2 .
-
Galamsey LTD has successfully acquired a license to mine in a 10,000-acre concession at the village of Franciskrom. Under the terms of the license, Galamsay is expected to restore the excavated land...
-
Convertible preferred stock is usually convertible to ________ at the ________ option. common stock; investor's a convertible bond; company's cash; company's a bond; investor's
-
Which concept states that only quantitative information can be recorded in books of accounts? Question 4 options: Separate Entity Concept Money Measurement Concept Accrual Concept Going Concern...
-
financial accounting and tax accounting are totally independent, so firms can simultaneously report high income to their shareholders while reporting low income to the IRS
-
Dr. Everton owns a dermatology practice. Approximately, twenty patients come through the facility a day. If every patient pays cash, what would the medical services be recorded as in double-entry...
-
Calculate the value of a call option on the stock in the previous problem with an exercise price of 110. Verify that the put-call parity theorem is satisfied by your answers to Problems 9 and 10. (Do...
-
A woman at a point A on the shore of a circular lake with radius 2 mi wants to arrive at the point C diametrically opposite on the other side of the lake in the shortest possible A time. She can walk...
-
Determine the pressure at the bottom of the tank in Fig. 3.25. 1.2 m --- --- -- -- Air 200 kPa (gage) Oil 15m/ (sg = 0.80) 2.6 m Water 2 m
-
Water is in the pipe shown in Fig. 3.26. Calculate the pressure at point A in kPa(gage). Pipe 100 mm - Water 75 mm Mercury (sg = 13.54)
-
For the differential manometer shown in Fig. 3.27, calculate the pressure difference between points A and B. The specific gravity of the oil is 0.85. 10 in 32 in Oil Water 9 in el
-
Example 6: Determine the resultant of the force system shown. Hint: use components method 6kips 15 7 8 10kips 6.8kips
-
The chart below shows images of 4 different samples taken through the two filters. Each column shows the same sample, but in a different filter. Sample 1 Sample 2 Sample 3 Sample 4 Filter 1 Filter 2...
-
(6 pts) 7) Setup (but do not evaluate) the integral you would use to find the total hydrostatic force on a circular plate with radius of 7m that is submerged vertically 2m below the surface of the...

Study smarter with the SolutionInn App


