Balance each of the following redox reactions in basic solution.
Question:
Balance each of the following redox reactions in basic solution.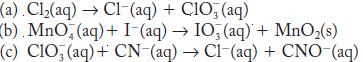
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To balance the redox reactions in a basic solution you need to follow several steps First identify the oxidation states of the elements and separate t...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Balance each of the following redox reactions in basic solution.
-
Balance each of the following redox reactions in basic solution.
-
Balance each of the following redox reactions in basic solution.
-
Suppose you are going to put up your own company, list down at least ten of your possible contributions to the socio-economic development in your community. Name of Business: Name of Owner/s: 2. 4....
-
The absorber plate of a solar collector may be coated with an opaque material for which the spectral, directional absorptivity is characterized by relations of the form the zenith angle is formed by...
-
A large number of consecutive IP address are available starting at 198.16.0.0. Suppose that four organizations, A, B, C, and D, request 4000, 2000, 4000, and 8000 addresses, respectively, and in that...
-
A brand-new sleeve bearing has been installed in a machine. The vibration levels on restart of the machine are not within tolerance, and the predominant peaks are at exactly one-half the operating...
-
Edney Manufacturing Company has $2 billion in sales and $0.6 billion in fixed assets. Currently, the companys fixed assets are operating at 80% of capacity. a. What level of sales could Edney have...
-
Selected current year-end financial statements of Cabot Corporation follow. (All sales were on credit; selected balance sheet amounts at December 31 of the prior year were inventory, $55,900; total...
-
Why is the following balanced reaction not a proper redox reaction?
-
Balance each of the following redox reactions in acid solution.
-
After identifying potential project risks, the project team should then ___________________. a. develop risk response plans for all identified risks. b. wait for the sponsor to conduct a risk...
-
Give three examples of digital media channels and briefly explain their communications benefits.
-
What are moments of truth?
-
Discuss the merits of the different models of paying for banner advertisements on the Internet for both media owners and companies placing advertisements.
-
Explain the implications of the Internet for Price.
-
What are meta tags? How important are they in ensuring a website is listed in a search engine?
-
The adjusted trial balance of Honeybee Hams, Inc., follows. Requirement Prepare Honeybee Hams, Inc.s income statement and statement of retained earnings for the year ended December 31, 2012, and its...
-
Rowland Textile Inc. manufactures two products: sweatshirts and T-shirts. The manufacturing process involves two activities: cutting and sewing. Expected overhead costs and cost drivers are as...
-
A squirrel is resting in a tall tree when it slips from a branch that is 50 m above the ground. It is a very agile squirrel and manages to land safely on another branch after only 0.50 s. What is the...
-
Basketball on the Moon. If LeBron James can jump 1.5 m high on Earth, how high could he jump on the Moon (assume an indoor court), where g - 1.6 m/s 2 ?
-
An apple falls from a branch near the top of a tall tree. If the branch is 12 m above the ground, what is the apples speed just before it hits the ground?
-
In 2023, Martinez receives the raw materials and pays the required $1,700,000. The raw materials now have a market value of $1,360,000. Prepare the entry to record the purchase. (Credit account...
-
How does the phenomenon of anticipatory anxiety manifest within the cognitive schema of stakeholders operating within highly evolved organizational ecosystems, and what strategies can be deployed to...
-
Delph Company uses a job - order costing system and has two manufacturing departments Molding and Fabrication. The company provided the following estimates at the beginning of the year: Molding...

Study smarter with the SolutionInn App


