Balance these reactions. (a) Al(s) + O(g) AlO3(s) (b) N(g) + H(g) NH3(g) (c) CH%(l)
Question:
Balance these reactions.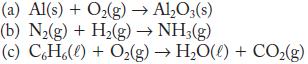
Transcribed Image Text:
(a) Al(s) + O₂(g) → Al₂O3(s) (b) N₂(g) + H₂(g) → NH3(g) (c) CH%(l) + Oz(g) → H,O(l) + CO,(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Lets balance the given chemical equations a Als O2g Al2O3s This eq...View the full answer

Answered By

Emily Grace
With over a decade of experience providing top-notch study assistance to students globally, I am dedicated to ensuring their academic success. My passion is to deliver original, high-quality assignments with fast turnaround times, always striving to exceed their expectations.
4.90+
3+ Reviews
22+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Balance these redox reactions by the half reaction method. a. Ca + H+ ( Ca2+ + H2 b. Sn2+ ( Sn + Sn4+ (Hint: both half reactions will start with the same reactant.)
-
Balance these redox reactions by the half reaction method. a. Fe3+ + Sn2+ ( Fe + Sn4+ b. Pb2+ ( Pb + Pb4+ (Hint: both half reactions will start with the same reactant.)
-
Balance these redox reactions by inspection. a. Na + F2 ( NaF b. Al2O3 + H2 ( Al + H2O
-
Describe the Economic Analysis of the Valero Energy Corporation. Demonstrate Valero Energy Corporation is susceptible to Macroeconomic outlook both in the U.S.& foreign markets
-
Three lossless lines are connected as shown in Figure. Determine Zin. 3A/4- SA/8 SA/8 /2- Z, = 75 2 z, - 100 2 Zia Z, - 60 - j 35 Z,= 502
-
By sketching their graphs or otherwise, find the range of these functions given the domain. State whether each function is a one-to-one function or a many-to-one function. a) f: x 2x + 1, x R b) g:...
-
Danielle Altman, the chief economist at Instawork, wrote an opinion column in the Wall Street Journal in which he described frictional unemployment as joblessness during the job-search process . . ....
-
Use the present value tables in the appendix on present value tables to calculate the issue price of a $300,000 bond issue in each of the following independent cases. Assume interest is paid...
-
QUESTION 3 (20 MKS) a) Obtain the time-independent Schrodinger Wave equation from the time dependent equation. [10 mks] b) Solve the time-independent Schrodinger equation given that,(x) = Aekx [10...
-
Presented below is a draft set of financial statements for Chips Limited. The following additional information is available: 1 Purchase invoices for goods received on 29 June 2010 amounting to...
-
(a) Write the equation for perchloric acid (HClO 4 ) dissolving in water. (b) Write the equation for sodium nitrate dissolving in water.
-
Write balanced equations for the following reactions. (a) NH4 + NO4N + HO (b) F + HO HF + O (c) NaO + HO NaOH
-
On October 1, 2010, the firm of Orson, Dorr, and Killough decided to liquidate their partnership. The partners have capital balances of $48,000, $63,000, and $11,000, respectively. The cash balance...
-
Explain the assumptions required for \(A R\) and ARDL forecasting.
-
Specify and estimate ARDL models. Use serial correlation checks, significance of coefficients, and model selection criteria to choose lag lengths.
-
For the simple regression model with heteroskedasticity, \(y_{i}=\beta_{1}+\beta_{2} x_{i}+e_{i}\) and \(\operatorname{var}\left(e_{i} \mid \mathbf{x}_{i} ight)=\sigma_{i}^{2}\) show that the...
-
Suppose that an outcome variable \(y_{i j}=\beta_{1}+\beta_{2} x_{i j}+e_{i j}, i=1, \ldots, N ; j=1, \ldots, N_{i}\). Assume \(E\left(e_{i j} \mid \mathbf{X} ight)=0\) and...
-
Use the internet search engine of your choice (Google, Bing, Yahoo!, etc.) to search for the top 10 best companies to work for. After looking over the rankings and explanations for each company, do...
-
Helmuth Inc's latest net income was $1,210,000, and it had 225,000 shares outstanding. The company wants to pay out 45% of its income. What dividend per share should it declare? a. $2.49 b. $2.06 c....
-
Michelles trust is subject to 3.8% surtax on the lesser of the trusts net investment income or the excess of the trusts adjusted gross income over the $12,400 threshold (the highest trust tax rate)....
-
You wish to design an effusion source for Br atoms from Br 2 (g). If the source is to operate at a total pressure of 7.5 Torr, what temperature is required to produce a degree of dissociation of...
-
Calculate G for the isothermal expansion of 2.25 mol of an ideal gas at 325 K from an initial pressure of 12.0 bar to a final pressure of 2.5 bar.
-
When ortho-bromonitrobenzene is treated with NaOH at elevated temperature, only one product is formed. (a) Draw the product. (b) Identify the intermediate formed en route to the product. (c) Would...
-
A small plastic ball of mass 5.86 10-3 kg and charge +0.178 C is suspended from an insulating thread and hangs between the plates of a capacitor (see the drawing). The ball is in equilibrium, with...
-
Assume that the firm invests $170,000 today and $200,000 three years from now to get $75,000 at Year 1, $80,000 at Year 2, $75,000 at Year 3, $140,000 at Year 4, $165,000 at Year 5, and $155,000 at...
-
During the first quarter of the year, Coronado Industries generated sales revenue of $1260000 on sales of 60000 units of its wireless earbuds. The break-even point is 30000 units. What is the...

Study smarter with the SolutionInn App


