Calculate the entropy change for the following processes.
Question:
Calculate the entropy change for the following processes.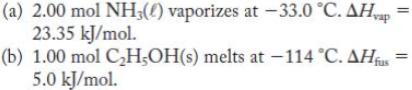
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To calculate the entropy change for the given processes we can use the formula for entropy change du...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Calculate the entropy change for the vaporization of liquid methane and hexane using the following data: Compare the molar volume of gaseous methane at 112 K with that of gaseous hexane at 342 K. How...
-
Calculate the entropy change for a process in which 3.00 moles of liquid water at 0oC is mixed with 1.00 mole of water at 100.oC in a perfectly insulated container. (Assume that the molar heat...
-
The accompanying diagram shows how entropy varies with temperature for a substance that is a gas at the highest temperature shown. (a) What processes correspond to the entropy increases along the...
-
Lacoste t-shirts come with an average price of $ 120 a piece, at their factory outlet with a std. deviation of $ 17. But at the Seasonal Sale (Discount) outlets of these t- shirts, it was also...
-
At tempted Grignard reaction of Cyclohexanone with tert-butyl magnesium bromide gives only about 1% yield of the expected addition product along with 99% un-reacted Cyclohexanone. If D 3 O + is added...
-
A pressure gage connected to a tank reads 500 kPa at a location where the atmospheric pressure is 94 kPa. Determine the absolute pressure in the tank.
-
The following are types of active surveillance except a. National household surveys gathering information on the social status and health status of people in a particular country b. Notifcation of...
-
Video Options Ltd. manufactures two types of DVD players: standard and deluxe. It attempts to set selling prices based on a 50% markup on manufacturing costs to cover selling and administrative...
-
A 2m rod of mass 8 kg is supported by a (zero-friction) pin-joint at A. It is released from rest from the horizontal position. When the bar reaches the position as shown in the diagram,=60. Answer...
-
Use data from Appendix G to calculate the standard entropy change for the following chemical reactions.
-
Calculate the entropy change for the following processes.
-
A compressed-air tank holds 0.500 m3 of air at a temperature of 295 K and a pressure of 820 kPa. What volume would the air occupy if it were released into the atmosphere, where the pressure is 101...
-
Describe an experience you have had with health literacy at the doctors office.
-
What is the difference between positioning and brand image?
-
Why is displaying prices so important in newspaper advertising?
-
Why is audience segmentation central to a social marketing approach in health promotion?
-
What other media could also be defined as retail advertising?
-
Answer these questions about two companies. 1. Emerald, Inc., began the year with total liabilities of $80,000 and total stockholders equity of $80,000. During the year, total assets increased by...
-
Write the binomial probability in words. Then, use a continuity correction to convert the binomial probability to a normal distribution probability. P(x 110)
-
Find R eq for the circuit in Fig. 2.94. 180 2 25 2 :60 2 60 2 Rea
-
All resistors (R) in Fig. 2.93 are 10 Ω each. Find R eq . R Reg R.
-
Design a problem, using Fig. 2.92 , to help other students better understand series and parallel circuits. Find v 1 , v 2 , and v 3 in the circuit in Fig. 2.92. + v1 - R3 v3 R2 v2
-
Vernon Company began operations on January 1 , year 1 , by issuing common stock for $ 3 1 , 0 0 0 cash. During year 1 , Vernon received $ 6 2 , 2 0 0 cash from revenue and incurred costs that...
-
The MegaMart Company began 2 0 2 4 with inventory of 1 9 , 0 0 0 units at a cost of $ 6 per unit. During 2 0 2 4 , 5 9 , 0 0 0 units were purchased for $ 7 . 2 0 each. Sales for the year totaled 6 7...
-
Based on the following sensitivity report, how much should the firm be willing to pay for 53 more units of Resource C? Variable Cells Cell $852 Name Product 1 Final Value Reduced Cost Objective...

Study smarter with the SolutionInn App


