Calculate the vapor pressure of each of the following at the given temperature. For each reaction, an
Question:
Calculate the vapor pressure of each of the following at the given temperature.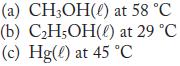
For each reaction, an equilibrium constant at 298 K is given. Calculate ΔG ° for each reaction.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Here are the calculated G values for each reaction ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Solve each formula for the specified variable. (Leave in the answers as needed.) P = kl 00 g for g
-
1. Explain why corrective action should be taken as soon as possible and practical following an infraction. 2. Why is it necessary to have a completely documented history leading up to an employee's...
-
Jin, the recruitment manager at Randents Inc., reviews the performance of his team members on a monthly basis. Based on the results of his monthly reviews, he decides to conduct daily reviews to...
-
For a prescribed wavelength A, measurement of the spectral intensity I.c (, T) = I, b of radiation emitted by a diffuse surface may be used to determine the surface temperature, if the spectral...
-
Distinguish between the terms electronegativity and electron affinity, covalent bond and ionic bond, and pure covalent bond and polar covalent bond. Characterize the types of bonds in terms of...
-
An observatory has a telescope that requires an initial investment of $\$ 25,000$. A benefactor will provide a $\$ 7,500$ gift every 5 years as long as the university makes the telescope available to...
-
Using Figure 7-5 as an example, redraw Figure 7-13 using an enterprise information system that processes a shared database. Explain the advantages of this system over the paper-based system in Figure...
-
Current Attempt in Progress The ledger of Cullumber, Inc. on March 31, 2027, includes the following selected accounts before adjusting entries. Debit Credit Prepaid Insurance $2,016 Supplies 2,100...
-
A 220-ft 3 sample of gas at standard temperature and pressure is compressed into a cylinder, where it exerts pressure of 2000 psi. Calculate the work (inJ) performed when this gas expands...
-
Calculate the vapor pressure of each of the following at the given temperature. For each reaction, an equilibrium constant at 298 K is given. Calculate G for each reaction.
-
What are the key tasks covered in the typical work program?
-
Joella is out shopping and finds a pair of sneakers originally valued at $96.00. They are now on sale for 75% of the original price. What is the new cost of the item? 2 answers
-
A high school has a total of 850 students. There are 60 more female students than there are male students. a. Write a system of linear equations that represents this situation. 2 answers
-
Isabel and Jake form a partnership. They want to build a new office. They get Larry, a real estate agent, to find them the perfect property. Isabel wants the partnership to have title to the new...
-
In what areas of the Filipino way of life did science contribute to nation-building during the pre-Hispanic Philippines?
-
1.Which year serves as the base year for the GDP Deflator? 2Real GDP for 1930 (expressed in terms of base-year dollars) is $_____ billion. 3.Real GDP for 1940 (expressed in terms of base-year...
-
How has the responsibility of auditors for planning and conducting the audit to detect fraud changed over the past 30 years?
-
In the busy port of Chennai, India, the number of containers loaded onto ships during a 15-week period is as follows: 1. Develop a linear trend equation to forecast container loadings. 2. Using the...
-
A large chipper/shredder is to be designed for use by commercial tree trimming companies. It would be mounted on a trailer to pull behind a large truck. The rotating blades of the unit protrude from...
-
A hot tub is to have 40 outlets that are each 8 mm in diameter with water exiting at 7 m/s. Treating each of the outlets as if they are at the surface of the water and exit into atmospheric pressure,...
-
A creek runs through a certain part of a campus where the water is falling about 2.5 m over a distance of just 8 m, and the creek before and after the fall is about 3 m wide. The sustainability club...
-
At the time that of its 10-Q filing of financial statements for the first half of its January 2002 fiscal year, Home Depots shares traded at $50 per share. The following are summaries from those...
-
121.Zen Arcade paid the weekly payroll on January 2 by debiting Salaries and Wages Expense for $47,000. The accountant preparing the payroll entry overlooked the fact that Salaries and Wages Expense...
-
Here is a plot of a vibrating string h: R2 R given by the C function h(x, t) for several times t. h(x,t) 0:5 1 - + 1.5 h(x,5) h(x, 4) = h(x,6) h(x, 3) = h(x,7) h(x,2) = h(x,8) h(x, 1) h(x,9) x 2

Study smarter with the SolutionInn App


