Concentrated nitric acid, HNO 3 , is 15.9 M and is frequently sold in 2.5-L containers .
Question:
Concentrated nitric acid, HNO3, is 15.9 M and is frequently sold in 2.5-L containers . How many moles of HNO3 are present in each container?
Strategy
Molarity, given in the problem, is the conversion between volume and moles present in a given volume of solution.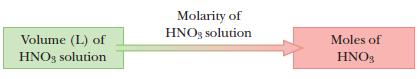
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:





