Find the equilibrium constant for the following reaction.
Question:
Find the equilibrium constant for the following reaction.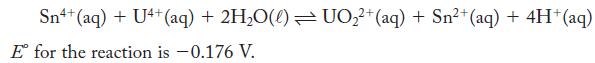
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Ke...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
The equilibrium constant for the following reaction is 1.0 Ã 10-3: Cr'. (aq) + H2EDTA2-(aq)--CrEDT A-(aq) + 2H' (aq) CH2-CO2 02GH CH EDTA N-CH,--CH2- O2C-CH2 CHy-CO2-...
-
Find the equilibrium constant for the reaction 2NO + O2 2NO2 from the elementary reactions in Table A.10 to answer which of the nitrogen oxides, NO or NO2; is the more stable at...
-
Find the equilibrium constant for the reaction 2NO + O2 2NO2 from the elementary reactions in Table A.11 to answer which of the nitrogen oxides, NO or NO2, is the more stable at ambient conditions?...
-
What characteristics do you think Jamie Oliver has that have helped make his various enterprises a success?
-
A horizontal semitransparent plate is uniformly irradiated from above and below, while air at T? = 300 K flows over the top and bottom surfaces, providing a uniform convection heat transfer...
-
Calculate the molar entropy of a constant-volume sample of argon at 250 K given that it is 154.84 J K-I mol-1 at 298 K.
-
A new pump installation is in process. The key fills the entire keyway in the pump shaft. That keyway is 2 inches long. The keyway in the pump coupling is 1 inch long. Does the key need to be cut,...
-
Preparing a consolidated balance sheet. The first two columns of Exhibit 13.13 present information from the accounting records of Ely Company and Sims Company at the end of the current year. Ely...
-
Harding Corp., a lamp manufacturer, provided the following information for the year ended December 31, 2024: (Click the icon to view account balances.) (Click the icon to view other information.)...
-
What is the voltage of the iron ion-copper cell when the [Fe 2+ ] = 1.55 M, [Fe 3+ ] 0.066 M, and [Cu 2+ ] = 0.500 M?
-
From the standard reduction potentials in Table 18.1, find the standard free energy change for the following reaction: Table 18.1
-
Do you believe the traditional or the Ricardian view of government debt? Why?
-
Describe the main features of internal check suitable for a bank.
-
Describe the procedure for audit of (a) Departmental undertakings and (b) Statutory corporations.
-
What are the registers to be maintained by an insurance company?
-
State how an auditor should outline the programme suitable for a share transfer audit.
-
How an auditor of a bank is appointed?
-
All-Star Advertising creates, plans, and handles advertising needs in the Tri-State area. Recently, All-Star had to replace an inexperienced office worker in charge of bookkeeping because of some...
-
What do you think?
-
Give an example in which the acceleration is perpendicular to the velocity.
-
For the system in Problem 12 and Figure P4.12, how large can m 2 be made without the system starting into motion? Data From Problem 12 Two blocks of mass m 1 = 45 kg and m 2 = 12 kg are connected by...
-
Give an example in which the magnitude of the instantaneous velocity is always larger than the average velocity.
-
In Skellig, when and how did reader find out that Skellig might be able to help Michael's baby sister? What do Skellig did when he disappeared from the tower?and who or what Skellig really was?
-
to calculate the amount of petrol sold for automobiles in mumbai in a month (30 days), we need to combine various pieces of information provided: 1) autos and buses in mumbai run on cng, so they do...
-
Can you elucidate the nuanced interdependencies between financial leverage, operational agility, and the propensity for organizational resurgence amidst the throes of decline within high-stakes,...

Study smarter with the SolutionInn App


